| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasmepsin X |
|---|
| Ligand | BDBM50591317 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2199854 (CHEMBL5112370) |
|---|
| IC50 | 7.0±n/a nM |
|---|
| Citation |  Lowe, MA; Cardenas, A; Valentin, JP; Zhu, Z; Abendroth, J; Castro, JL; Class, R; Delaunois, A; Fleurance, R; Gerets, H; Gryshkova, V; King, L; Lorimer, DD; MacCoss, M; Rowley, JH; Rosseels, ML; Royer, L; Taylor, RD; Wong, M; Zaccheo, O; Chavan, VP; Ghule, GA; Tapkir, BK; Burrows, JN; Duffey, M; Rottmann, M; Wittlin, S; Angulo-Barturen, I; Jim�nez-D�az, MB; Striepen, J; Fairhurst, KJ; Yeo, T; Fidock, DA; Cowman, AF; Favuzza, P; Crespo-Fernandez, B; Gamo, FJ; Goldberg, DE; Soldati-Favre, D; Laleu, B; de Haro, T Discovery and Characterization of Potent, Efficacious, Orally Available Antimalarial Plasmepsin X Inhibitors and Preclinical Safety Assessment of J Med Chem65:14121-14143 (2022) [PubMed] Article Lowe, MA; Cardenas, A; Valentin, JP; Zhu, Z; Abendroth, J; Castro, JL; Class, R; Delaunois, A; Fleurance, R; Gerets, H; Gryshkova, V; King, L; Lorimer, DD; MacCoss, M; Rowley, JH; Rosseels, ML; Royer, L; Taylor, RD; Wong, M; Zaccheo, O; Chavan, VP; Ghule, GA; Tapkir, BK; Burrows, JN; Duffey, M; Rottmann, M; Wittlin, S; Angulo-Barturen, I; Jim�nez-D�az, MB; Striepen, J; Fairhurst, KJ; Yeo, T; Fidock, DA; Cowman, AF; Favuzza, P; Crespo-Fernandez, B; Gamo, FJ; Goldberg, DE; Soldati-Favre, D; Laleu, B; de Haro, T Discovery and Characterization of Potent, Efficacious, Orally Available Antimalarial Plasmepsin X Inhibitors and Preclinical Safety Assessment of J Med Chem65:14121-14143 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Plasmepsin X |
|---|
| Name: | Plasmepsin X |
|---|
| Synonyms: | 3.4.23.- | PLM10_PLAF7 | PMX | PfPMX | Plasmepsin 10 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65102.67 |
|---|
| Organism: | Plasmodium falciparum (isolate 3D7) |
|---|
| Description: | ChEMBL_119140 |
|---|
| Residue: | 573 |
|---|
| Sequence: | MKRISPLNTLFYLSLFFSYTFKGLKCTRIYKIGTKALPCSECHDVFDCTGCLFEEKESSH
VIPLKLNKKNPNDHKKLQKHHESLKLGDVKYYVNRGEGISGSLGTSSGNTLDDMDLINEE
INKKRTNAQLDEKNFLDFTTYNKNKAQDISDHLSDIQKHVYEQDAQKGNKNFTNNENNSD
NENNSDNENNSDNENNLDNENNLDNENNSDNSSIEKNFIALENKNATVEQTKENIFLVPL
KHLRDSQFVGELLVGTPPQTVYPIFDTGSTNVWVVTTACEEESCKKVRRYDPNKSKTFRR
SFIEKNLHIVFGSGSISGSVGTDTFMLGKHLVRNQTFGLVESESNNNKNGGDNIFDYISF
EGIVGLGFPGMLSAGNIPFFDNLLKQNPNVDPQFSFYISPYDGKSTLIIGGISKSFYEGD
IYMLPVLKESYWEVKLDELYIGKERICCDEESYVIFDTGTSYNTMPSSQMKTFLNLIHST
ACTEQNYKDILKSYPIIKYVFGELIIELHPEEYMILNDDVCMPAYMQIDVPSERNHAYLL
GSLSFMRNFFTVFVRGTESRPSMVGVARAKSKN
|
|
|
|---|
| BDBM50591317 |
|---|
| n/a |
|---|
| Name | BDBM50591317 |
|---|
| Synonyms: | CHEMBL5191783 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H30ClN5O2 |
|---|
| Mol. Mass. | 467.991 |
|---|
| SMILES | C[C@H]1C[C@H](CCO1)N1C(=O)C[C@](C)(NC1=N)c1cccc(Nc2ccc(nc2)C2CC2)c1Cl |r| |
|---|
| Structure | 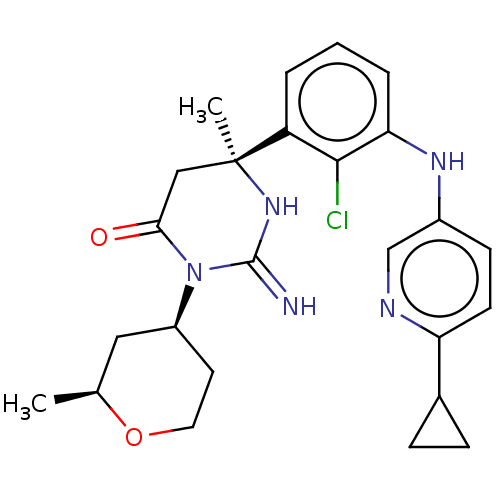
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lowe, MA; Cardenas, A; Valentin, JP; Zhu, Z; Abendroth, J; Castro, JL; Class, R; Delaunois, A; Fleurance, R; Gerets, H; Gryshkova, V; King, L; Lorimer, DD; MacCoss, M; Rowley, JH; Rosseels, ML; Royer, L; Taylor, RD; Wong, M; Zaccheo, O; Chavan, VP; Ghule, GA; Tapkir, BK; Burrows, JN; Duffey, M; Rottmann, M; Wittlin, S; Angulo-Barturen, I; Jim�nez-D�az, MB; Striepen, J; Fairhurst, KJ; Yeo, T; Fidock, DA; Cowman, AF; Favuzza, P; Crespo-Fernandez, B; Gamo, FJ; Goldberg, DE; Soldati-Favre, D; Laleu, B; de Haro, T Discovery and Characterization of Potent, Efficacious, Orally Available Antimalarial Plasmepsin X Inhibitors and Preclinical Safety Assessment of J Med Chem65:14121-14143 (2022) [PubMed] Article
Lowe, MA; Cardenas, A; Valentin, JP; Zhu, Z; Abendroth, J; Castro, JL; Class, R; Delaunois, A; Fleurance, R; Gerets, H; Gryshkova, V; King, L; Lorimer, DD; MacCoss, M; Rowley, JH; Rosseels, ML; Royer, L; Taylor, RD; Wong, M; Zaccheo, O; Chavan, VP; Ghule, GA; Tapkir, BK; Burrows, JN; Duffey, M; Rottmann, M; Wittlin, S; Angulo-Barturen, I; Jim�nez-D�az, MB; Striepen, J; Fairhurst, KJ; Yeo, T; Fidock, DA; Cowman, AF; Favuzza, P; Crespo-Fernandez, B; Gamo, FJ; Goldberg, DE; Soldati-Favre, D; Laleu, B; de Haro, T Discovery and Characterization of Potent, Efficacious, Orally Available Antimalarial Plasmepsin X Inhibitors and Preclinical Safety Assessment of J Med Chem65:14121-14143 (2022) [PubMed] Article