| Reaction Details |
|---|
 | Report a problem with these data |
| Target | L-lactate dehydrogenase B chain |
|---|
| Ligand | BDBM50594014 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2210642 (CHEMBL5123591) |
|---|
| IC50 | 1100±n/a nM |
|---|
| Citation |  Moya-Garzon, MD; Rodriguez-Rodriguez, B; Martin-Higueras, C; Franco-Montalban, F; Fernandes, MX; Gomez-Vidal, JA; Pey, AL; Salido, E; Diaz-Gavilan, M New salicylic acid derivatives, double inhibitors of glycolate oxidase and lactate dehydrogenase, as effective agents decreasing oxalate production. Eur J Med Chem237:0 (2022) [PubMed] Article Moya-Garzon, MD; Rodriguez-Rodriguez, B; Martin-Higueras, C; Franco-Montalban, F; Fernandes, MX; Gomez-Vidal, JA; Pey, AL; Salido, E; Diaz-Gavilan, M New salicylic acid derivatives, double inhibitors of glycolate oxidase and lactate dehydrogenase, as effective agents decreasing oxalate production. Eur J Med Chem237:0 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| L-lactate dehydrogenase B chain |
|---|
| Name: | L-lactate dehydrogenase B chain |
|---|
| Synonyms: | L-lactate dehydrogenase B | L-lactate dehydrogenase B chain | LDH heart subunit | LDH-B | LDH-H | LDHB | LDHB_HUMAN | Lactate dehydrogenase B (LDHB) | Renal carcinoma antigen NY-REN-46 |
|---|
| Type: | Oxidoreductase; homotetramer |
|---|
| Mol. Mass.: | 36635.55 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 334 |
|---|
| Sequence: | MATLKEKLIAPVAEEEATVPNNKITVVGVGQVGMACAISILGKSLADELALVDVLEDKLK
GEMMDLQHGSLFLQTPKIVADKDYSVTANSKIVVVTAGVRQQEGESRLNLVQRNVNVFKF
IIPQIVKYSPDCIIIVVSNPVDILTYVTWKLSGLPKHRVIGSGCNLDSARFRYLMAEKLG
IHPSSCHGWILGEHGDSSVAVWSGVNVAGVSLQELNPEMGTDNDSENWKEVHKMVVESAY
EVIKLKGYTNWAIGLSVADLIESMLKNLSRIHPVSTMVKGMYGIENEVFLSLPCILNARG
LTSVINQKLKDDEVAQLKKSADTLWDIQKDLKDL
|
|
|
|---|
| BDBM50594014 |
|---|
| n/a |
|---|
| Name | BDBM50594014 |
|---|
| Synonyms: | CHEMBL5182027 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H17BrN2O4 |
|---|
| Mol. Mass. | 405.243 |
|---|
| SMILES | N.OC(=O)c1cc(ccc1O)-c1ccc(CNc2ccc(Br)cc2)o1 |
|---|
| Structure | 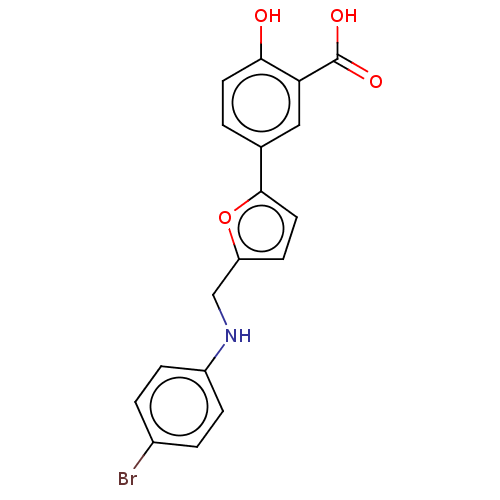
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Moya-Garzon, MD; Rodriguez-Rodriguez, B; Martin-Higueras, C; Franco-Montalban, F; Fernandes, MX; Gomez-Vidal, JA; Pey, AL; Salido, E; Diaz-Gavilan, M New salicylic acid derivatives, double inhibitors of glycolate oxidase and lactate dehydrogenase, as effective agents decreasing oxalate production. Eur J Med Chem237:0 (2022) [PubMed] Article
Moya-Garzon, MD; Rodriguez-Rodriguez, B; Martin-Higueras, C; Franco-Montalban, F; Fernandes, MX; Gomez-Vidal, JA; Pey, AL; Salido, E; Diaz-Gavilan, M New salicylic acid derivatives, double inhibitors of glycolate oxidase and lactate dehydrogenase, as effective agents decreasing oxalate production. Eur J Med Chem237:0 (2022) [PubMed] Article