| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50169301 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302654 (CHEMBL839943) |
|---|
| Ki | 5±n/a nM |
|---|
| Citation |  Matasi, JJ; Caldwell, JP; Zhang, H; Fawzi, A; Cohen-Williams, ME; Varty, GB; Tulshian, DB 2-(2-Furanyl)-7-phenyl[1,2,4]triazolo[1,5-c]pyrimidin-5-amine analogs: highly potent, orally active, adenosine A2A antagonists. Part 1. Bioorg Med Chem Lett15:3670-4 (2005) [PubMed] Article Matasi, JJ; Caldwell, JP; Zhang, H; Fawzi, A; Cohen-Williams, ME; Varty, GB; Tulshian, DB 2-(2-Furanyl)-7-phenyl[1,2,4]triazolo[1,5-c]pyrimidin-5-amine analogs: highly potent, orally active, adenosine A2A antagonists. Part 1. Bioorg Med Chem Lett15:3670-4 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | AA2AR_RAT | ADENOSINE A2a | Adenosine A2 receptor | Adenosine A2a receptor (A2a) | Adenosine Receptors A2a (A2a) | Adenosine receptor A2a and A3 | Adenosine receptors A2a | Adora2a | Rat striatal adenosine A2a receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 45015.65 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat A2A receptors expressed in CHO cells. |
|---|
| Residue: | 410 |
|---|
| Sequence: | MGSSVYITVELAIAVLAILGNVLVCWAVWINSNLQNVTNFFVVSLAAADIAVGVLAIPFA
ITISTGFCAACHGCLFFACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGVRAKG
IIAICWVLSFAIGLTPMLGWNNCSQKDGNSTKTCGEGRVTCLFEDVVPMNYMVYYNFFAF
VLLPLLLMLAIYLRIFLAARRQLKQMESQPLPGERTRSTLQKEVHAAKSLAIIVGLFALC
WLPLHIINCFTFFCSTCRHAPPWLMYLAIILSHSNSVVNPFIYAYRIREFRQTFRKIIRT
HVLRRQEPFQAGGSSAWALAAHSTEGEQVSLRLNGHPLGVWANGSATHSGRRPNGYTLGL
GGGGSAQGSPRDVELPTQERQEGQEHPGLRGHLVQARVGASSWSSEFAPS
|
|
|
|---|
| BDBM50169301 |
|---|
| n/a |
|---|
| Name | BDBM50169301 |
|---|
| Synonyms: | 7-(3-Amino-phenyl)-2-furan-2-yl-[1,2,4]triazolo[1,5-c]pyrimidin-5-ylamine | CHEMBL366905 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H12N6O |
|---|
| Mol. Mass. | 292.2954 |
|---|
| SMILES | Nc1cccc(c1)-c1cc2nc(nn2c(N)n1)-c1ccco1 |
|---|
| Structure | 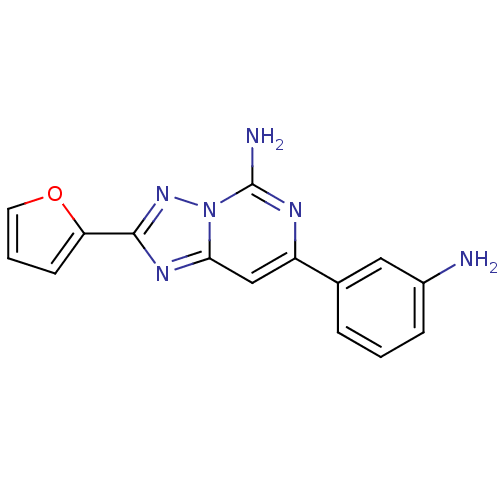
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Matasi, JJ; Caldwell, JP; Zhang, H; Fawzi, A; Cohen-Williams, ME; Varty, GB; Tulshian, DB 2-(2-Furanyl)-7-phenyl[1,2,4]triazolo[1,5-c]pyrimidin-5-amine analogs: highly potent, orally active, adenosine A2A antagonists. Part 1. Bioorg Med Chem Lett15:3670-4 (2005) [PubMed] Article
Matasi, JJ; Caldwell, JP; Zhang, H; Fawzi, A; Cohen-Williams, ME; Varty, GB; Tulshian, DB 2-(2-Furanyl)-7-phenyl[1,2,4]triazolo[1,5-c]pyrimidin-5-amine analogs: highly potent, orally active, adenosine A2A antagonists. Part 1. Bioorg Med Chem Lett15:3670-4 (2005) [PubMed] Article