| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Rho-associated protein kinase 2 |
|---|
| Ligand | BDBM50183844 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_354929 (CHEMBL870120) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Hennequin, LF; Ballard, P; Boyle, FT; Delouvrié, B; Ellston, RP; Halsall, CT; Harris, CS; Hudson, K; Kendrew, J; Pease, JE; Ross, HS; Smith, P; Vincent, JL Novel 4-anilinoquinazolines with C-6 carbon-linked side chains: synthesis and structure-activity relationship of a series of potent, orally active, EGF receptor tyrosine kinase inhibitors. Bioorg Med Chem Lett16:2672-6 (2006) [PubMed] Article Hennequin, LF; Ballard, P; Boyle, FT; Delouvrié, B; Ellston, RP; Halsall, CT; Harris, CS; Hudson, K; Kendrew, J; Pease, JE; Ross, HS; Smith, P; Vincent, JL Novel 4-anilinoquinazolines with C-6 carbon-linked side chains: synthesis and structure-activity relationship of a series of potent, orally active, EGF receptor tyrosine kinase inhibitors. Bioorg Med Chem Lett16:2672-6 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Rho-associated protein kinase 2 |
|---|
| Name: | Rho-associated protein kinase 2 |
|---|
| Synonyms: | KIAA0619 | ROCK-II | ROCK2 | ROCK2_HUMAN | Rho kinase 2 (ROCKII) | Rho-associated protein kinase 2 (ROCK-2) | Rho-associated protein kinase 2 (ROCK2) | Rho-associated protein kinase 2 (ROCKII) | Rho-associated protein kinase 2/Transforming protein RhoA | Rho-associated, coiled-coil-containing protein kinase 2 | Rho-associated, coiled-coil-containing protein kinase II | Serine/threonine-protein kinase RIO2 | p164 ROCK-2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 160885.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O75116 |
|---|
| Residue: | 1388 |
|---|
| Sequence: | MSRPPPTGKMPGAPETAPGDGAGASRQRKLEALIRDPRSPINVESLLDGLNSLVLDLDFP
ALRKNKNIDNFLNRYEKIVKKIRGLQMKAEDYDVVKVIGRGAFGEVQLVRHKASQKVYAM
KLLSKFEMIKRSDSAFFWEERDIMAFANSPWVVQLFYAFQDDRYLYMVMEYMPGGDLVNL
MSNYDVPEKWAKFYTAEVVLALDAIHSMGLIHRDVKPDNMLLDKHGHLKLADFGTCMKMD
ETGMVHCDTAVGTPDYISPEVLKSQGGDGFYGRECDWWSVGVFLYEMLVGDTPFYADSLV
GTYSKIMDHKNSLCFPEDAEISKHAKNLICAFLTDREVRLGRNGVEEIRQHPFFKNDQWH
WDNIRETAAPVVPELSSDIDSSNFDDIEDDKGDVETFPIPKAFVGNQLPFIGFTYYRENL
LLSDSPSCRETDSIQSRKNEESQEIQKKLYTLEEHLSNEMQAKEELEQKCKSVNTRLEKT
AKELEEEITLRKSVESALRQLEREKALLQHKNAEYQRKADHEADKKRNLENDVNSLKDQL
EDLKKRNQNSQISTEKVNQLQRQLDETNALLRTESDTAARLRKTQAESSKQIQQLESNNR
DLQDKNCLLETAKLKLEKEFINLQSALESERRDRTHGSEIINDLQGRICGLEEDLKNGKI
LLAKVELEKRQLQERFTDLEKEKSNMEIDMTYQLKVIQQSLEQEEAEHKATKARLADKNK
IYESIEEAKSEAMKEMEKKLLEERTLKQKVENLLLEAEKRCSLLDCDLKQSQQKINELLK
QKDVLNEDVRNLTLKIEQETQKRCLTQNDLKMQTQQVNTLKMSEKQLKQENNHLMEMKMN
LEKQNAELRKERQDADGQMKELQDQLEAEQYFSTLYKTQVRELKEECEEKTKLGKELQQK
KQELQDERDSLAAQLEITLTKADSEQLARSIAEEQYSDLEKEKIMKELEIKEMMARHKQE
LTEKDATIASLEETNRTLTSDVANLANEKEELNNKLKDVQEQLSRLKDEEISAAAIKAQF
EKQLLTERTLKTQAVNKLAEIMNRKEPVKRGNDTDVRRKEKENRKLHMELKSEREKLTQQ
MIKYQKELNEMQAQIAEESQIRIELQMTLDSKDSDIEQLRSQLQALHIGLDSSSIGSGPG
DAEADDGFPESRLEGWLSLPVRNNTKKFGWVKKYVIVSSKKILFYDSEQDKEQSNPYMVL
DIDKLFHVRPVTQTDVYRADAKEIPRIFQILYANEGESKKEQEFPVEPVGEKSNYICHKG
HEFIPTLYHFPTNCEACMKPLWHMFKPPPALECRRCHIKCHKDHMDKKEEIIAPCKVYYD
ISTAKNLLLLANSTEEQQKWVSRLVKKIPKKPPAPDPFARSSPRTSMKIQQNQSIRRPSR
QLAPNKPS
|
|
|
|---|
| BDBM50183844 |
|---|
| n/a |
|---|
| Name | BDBM50183844 |
|---|
| Synonyms: | (S)-2-(((4-(3-chloro-2-fluorophenylamino)-7-methoxyquinazolin-6-yl)methyl)(methyl)amino)-3-hydroxypropanamide | CHEMBL208286 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21ClFN5O3 |
|---|
| Mol. Mass. | 433.864 |
|---|
| SMILES | COc1cc2ncnc(Nc3cccc(Cl)c3F)c2cc1CN(C)[C@@H](CO)C(N)=O |
|---|
| Structure | 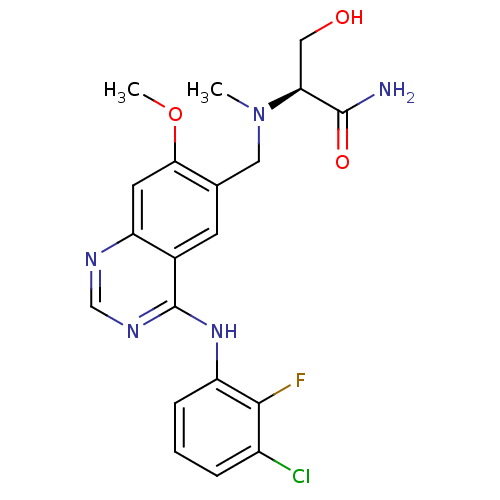
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hennequin, LF; Ballard, P; Boyle, FT; Delouvrié, B; Ellston, RP; Halsall, CT; Harris, CS; Hudson, K; Kendrew, J; Pease, JE; Ross, HS; Smith, P; Vincent, JL Novel 4-anilinoquinazolines with C-6 carbon-linked side chains: synthesis and structure-activity relationship of a series of potent, orally active, EGF receptor tyrosine kinase inhibitors. Bioorg Med Chem Lett16:2672-6 (2006) [PubMed] Article
Hennequin, LF; Ballard, P; Boyle, FT; Delouvrié, B; Ellston, RP; Halsall, CT; Harris, CS; Hudson, K; Kendrew, J; Pease, JE; Ross, HS; Smith, P; Vincent, JL Novel 4-anilinoquinazolines with C-6 carbon-linked side chains: synthesis and structure-activity relationship of a series of potent, orally active, EGF receptor tyrosine kinase inhibitors. Bioorg Med Chem Lett16:2672-6 (2006) [PubMed] Article