| Reaction Details |
|---|
 | Report a problem with these data |
| Target | GTPase KRas |
|---|
| Ligand | BDBM50604229 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2247236 (CHEMBL5161446) |
|---|
| Kd | 33±n/a nM |
|---|
| Citation |  Li, L; Liu, J; Yang, Z; Zhao, H; Deng, B; Ren, Y; Mai, R; Huang, J; Chen, J Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening. Eur J Med Chem233:0 (2022) [PubMed] Article Li, L; Liu, J; Yang, Z; Zhao, H; Deng, B; Ren, Y; Mai, R; Huang, J; Chen, J Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening. Eur J Med Chem233:0 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| GTPase KRas |
|---|
| Name: | GTPase KRas |
|---|
| Synonyms: | GTPase KRas, N-terminally processed | K-Ras 2 | KRAS | KRAS2 | Ki-Ras | RASK2 | RASK_HUMAN | c-K-ras | c-Ki-ras |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 21656.10 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1476955 |
|---|
| Residue: | 189 |
|---|
| Sequence: | MTEYKLVVVGAGGVGKSALTIQLIQNHFVDEYDPTIEDSYRKQVVIDGETCLLDILDTAG
QEEYSAMRDQYMRTGEGFLCVFAINNTKSFEDIHHYREQIKRVKDSEDVPMVLVGNKCDL
PSRTVDTKQAQDLARSYGIPFIETSAKTRQRVEDAFYTLVREIRQYRLKKISKEEKTPGC
VKIKKCIIM
|
|
|
|---|
| BDBM50604229 |
|---|
| n/a |
|---|
| Name | BDBM50604229 |
|---|
| Synonyms: | CHEMBL5205540 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H19F3N4OS |
|---|
| Mol. Mass. | 420.451 |
|---|
| SMILES | COc1ccc(cc1-c1cc2c(ncnc2s1)N1CC2CCC(C1)N2)C(F)(F)F |TLB:11:17:24:20.21| |
|---|
| Structure | 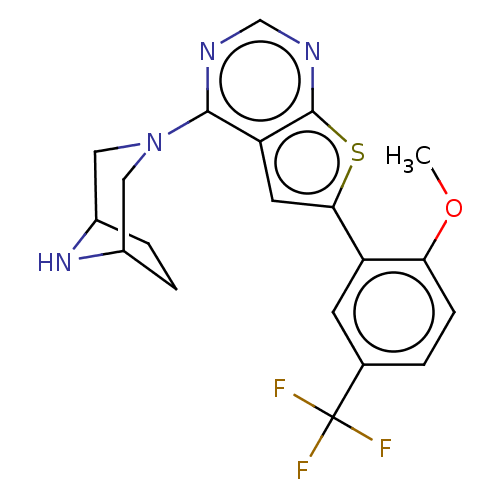
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, L; Liu, J; Yang, Z; Zhao, H; Deng, B; Ren, Y; Mai, R; Huang, J; Chen, J Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening. Eur J Med Chem233:0 (2022) [PubMed] Article
Li, L; Liu, J; Yang, Z; Zhao, H; Deng, B; Ren, Y; Mai, R; Huang, J; Chen, J Discovery of Thieno[2,3-d]pyrimidine-based KRAS G12D inhibitors as potential anticancer agents via combinatorial virtual screening. Eur J Med Chem233:0 (2022) [PubMed] Article