| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 17-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Ligand | BDBM50188390 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_362612 (CHEMBL870159) |
|---|
| IC50 | 28300±n/a nM |
|---|
| Citation |  Schuster, D; Maurer, EM; Laggner, C; Nashev, LG; Wilckens, T; Langer, T; Odermatt, A The discovery of new 11beta-hydroxysteroid dehydrogenase type 1 inhibitors by common feature pharmacophore modeling and virtual screening. J Med Chem49:3454-66 (2006) [PubMed] Article Schuster, D; Maurer, EM; Laggner, C; Nashev, LG; Wilckens, T; Langer, T; Odermatt, A The discovery of new 11beta-hydroxysteroid dehydrogenase type 1 inhibitors by common feature pharmacophore modeling and virtual screening. J Med Chem49:3454-66 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 17-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Name: | 17-beta-hydroxysteroid dehydrogenase type 2 |
|---|
| Synonyms: | 17-beta-Hydroxysteroid Dehydrogenase 2 (17-beta-HSD2) | 20 alpha-hydroxysteroid dehydrogenase | 20-alpha-HSD | DHB2_HUMAN | EDH17B2 | Estradiol 17-beta-dehydrogenase 2 | Estradiol 17-beta-dehydrogenase 2 (17beta-HSD2) | HSD17B2 | Microsomal 17-beta-hydroxysteroid dehydrogenas | SDR9C2 | Testosterone 17-beta-dehydrogenase |
|---|
| Type: | Oxidoreductase; single-pass type II membrane protein |
|---|
| Mol. Mass.: | 42795.29 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Enzyme was partially purified from human placenta. |
|---|
| Residue: | 387 |
|---|
| Sequence: | MSTFFSDTAWICLAVPTVLCGTVFCKYKKSSGQLWSWMVCLAGLCAVCLLILSPFWGLIL
FSVSCFLMYTYLSGQELLPVDQKAVLVTGGDCGLGHALCKYLDELGFTVFAGVLNENGPG
AEELRRTCSPRLSVLQMDITKPVQIKDAYSKVAAMLQDRGLWAVINNAGVLGFPTDGELL
LMTDYKQCMAVNFFGTVEVTKTFLPLLRKSKGRLVNVSSMGGGAPMERLASYGSSKAAVT
MFSSVMRLELSKWGIKVASIQPGGFLTNIAGTSDKWEKLEKDILDHLPAEVQEDYGQDYI
LAQRNFLLLINSLASKDFSPVLRDIQHAILAKSPFAYYTPGKGAYLWICLAHYLPIGIYD
YFAKRHFGQDKPMPRALRMPNYKKKAT
|
|
|
|---|
| BDBM50188390 |
|---|
| n/a |
|---|
| Name | BDBM50188390 |
|---|
| Synonyms: | 4-((5R,10S,13R,14R)-3-acetoxy-4,4,10,13,14-pentamethyl-7,11-dioxo-2,3,4,5,6,7,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl)pentanoic acid | CHEMBL211039 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H42O6 |
|---|
| Mol. Mass. | 486.6402 |
|---|
| SMILES | CC(CCC(O)=O)C1CC[C@@]2(C)C3=C(C(=O)C[C@]12C)[C@@]1(C)CCC(OC(C)=O)C(C)(C)[C@@H]1CC3=O |c:12| |
|---|
| Structure | 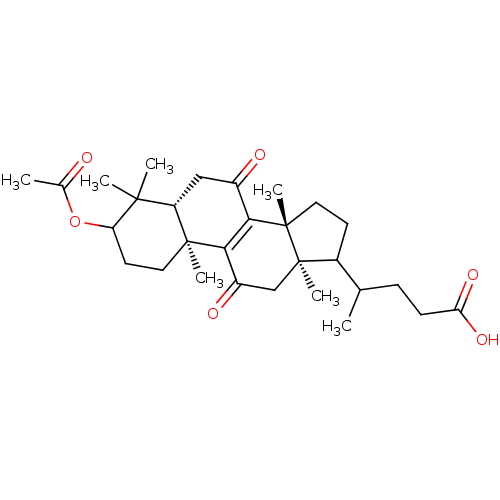
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Schuster, D; Maurer, EM; Laggner, C; Nashev, LG; Wilckens, T; Langer, T; Odermatt, A The discovery of new 11beta-hydroxysteroid dehydrogenase type 1 inhibitors by common feature pharmacophore modeling and virtual screening. J Med Chem49:3454-66 (2006) [PubMed] Article
Schuster, D; Maurer, EM; Laggner, C; Nashev, LG; Wilckens, T; Langer, T; Odermatt, A The discovery of new 11beta-hydroxysteroid dehydrogenase type 1 inhibitors by common feature pharmacophore modeling and virtual screening. J Med Chem49:3454-66 (2006) [PubMed] Article