| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Transporter |
|---|
| Ligand | BDBM22165 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_372239 (CHEMBL853393) |
|---|
| Ki | 102±n/a nM |
|---|
| Citation |  Zhang, S; Fernandez, F; Hazeldine, S; Deschamps, J; Zhen, J; Reith, ME; Dutta, AK Further structural exploration of trisubstituted asymmetric pyran derivatives (2S,4R,5R)-2-benzhydryl-5-benzylamino-tetrahydropyran-4-ol and their corresponding disubstituted (3S,6S) pyran derivatives: a proposed pharmacophore model for high-affinity interaction with the dopamine, serotonin, and no J Med Chem49:4239-47 (2006) [PubMed] Article Zhang, S; Fernandez, F; Hazeldine, S; Deschamps, J; Zhen, J; Reith, ME; Dutta, AK Further structural exploration of trisubstituted asymmetric pyran derivatives (2S,4R,5R)-2-benzhydryl-5-benzylamino-tetrahydropyran-4-ol and their corresponding disubstituted (3S,6S) pyran derivatives: a proposed pharmacophore model for high-affinity interaction with the dopamine, serotonin, and no J Med Chem49:4239-47 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Transporter |
|---|
| Name: | Transporter |
|---|
| Synonyms: | Monoamine transporters; Norepininephrine & dopamine | Norepinephrine Monoamine transporters |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 66787.87 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1463061 |
|---|
| Residue: | 597 |
|---|
| Sequence: | MVTRTRETWGKKIDFLLSVVGFAVDLANVWRFPYLCYKNGGGAFLIPYTLFLIIAGMPLF
YMELALGQFNREGAATVWKICPFFKGVGYAVILIALYVGFYYNVIIAWSLYYLFASFTLN
LPWTNCGHAWNSPNCTDPKLLNASVLGDHTKYSKYKFTPAAEFYERGVLHLHESSGIHDI
GLPQWQLLLCLMVVIVVLYVSLWKGVKTSGKVVWITATLPYFVLFVLLVHGVTLPGASNG
INAYLHIDFYRLKEATVWIDAATQIFFSLGAGFGVLIAFASYNKFDNNCYRDALLTSTIN
CVTSFISGFAIFSILGYMAHEHKVKIEDVATEGAGLVFVLYPEAISTLSGSTFWAVLFFL
MLLALGLDSSMGGMEAVITGLADDFQVLKRHRKLFTCAVTLGTFLLAMFCITKGGIYVLT
LLDTFAAGTSILFAVLMEAIGVSWFYGVDRFSNDIQQMMGFKPGLYWRLCWKFVSPAFLL
FVVVVSIINFKPLTYDDYVYPPWANWVGWGIALSSMILVPAYVIYKFFSIRGSLWERVAY
GITPENEHHLLALEIELSSLQSFVITSCPIDPLLSSFLFISCQKTLVFKKSGPAPLI
|
|
|
|---|
| BDBM22165 |
|---|
| n/a |
|---|
| Name | BDBM22165 |
|---|
| Synonyms: | 1-{2-[bis(4-fluorophenyl)methoxy]ethyl}-4-(3-phenylpropyl)piperazine | CHEMBL153260 | CHEMBL281594 | CHEMBL542933 | CHEMBL543876 | GBR 12909 | GBR-12909 | NCGC00015300 | US9944618, Compound ID No. 175 | Vanoxerine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H32F2N2O |
|---|
| Mol. Mass. | 450.5633 |
|---|
| SMILES | Fc1ccc(cc1)C(OCCN1CCN(CCCc2ccccc2)CC1)c1ccc(F)cc1 |
|---|
| Structure | 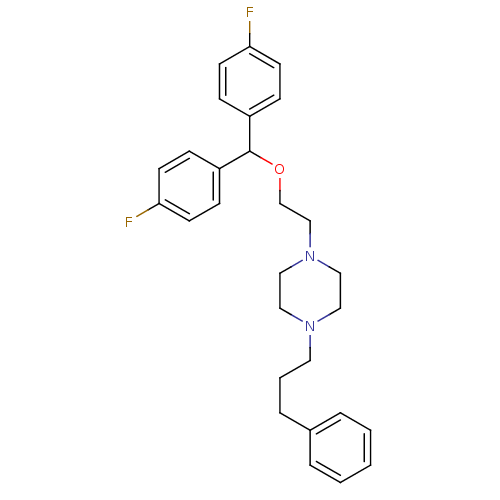
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, S; Fernandez, F; Hazeldine, S; Deschamps, J; Zhen, J; Reith, ME; Dutta, AK Further structural exploration of trisubstituted asymmetric pyran derivatives (2S,4R,5R)-2-benzhydryl-5-benzylamino-tetrahydropyran-4-ol and their corresponding disubstituted (3S,6S) pyran derivatives: a proposed pharmacophore model for high-affinity interaction with the dopamine, serotonin, and no J Med Chem49:4239-47 (2006) [PubMed] Article
Zhang, S; Fernandez, F; Hazeldine, S; Deschamps, J; Zhen, J; Reith, ME; Dutta, AK Further structural exploration of trisubstituted asymmetric pyran derivatives (2S,4R,5R)-2-benzhydryl-5-benzylamino-tetrahydropyran-4-ol and their corresponding disubstituted (3S,6S) pyran derivatives: a proposed pharmacophore model for high-affinity interaction with the dopamine, serotonin, and no J Med Chem49:4239-47 (2006) [PubMed] Article