| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase Yes |
|---|
| Ligand | BDBM50193874 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_424209 (CHEMBL908983) |
|---|
| IC50 | 1±n/a nM |
|---|
| Citation |  Noronha, G; Barrett, K; Cao, J; Dneprovskaia, E; Fine, R; Gong, X; Gritzen, C; Hood, J; Kang, X; Klebansky, B; Li, G; Liao, W; Lohse, D; Mak, CC; McPherson, A; Palanki, MS; Pathak, VP; Renick, J; Soll, R; Splittgerber, U; Wrasidlo, W; Zeng, B; Zhao, N; Zhou, Y Discovery and preliminary structure-activity relationship studies of novel benzotriazine based compounds as Src inhibitors. Bioorg Med Chem Lett16:5546-50 (2006) [PubMed] Article Noronha, G; Barrett, K; Cao, J; Dneprovskaia, E; Fine, R; Gong, X; Gritzen, C; Hood, J; Kang, X; Klebansky, B; Li, G; Liao, W; Lohse, D; Mak, CC; McPherson, A; Palanki, MS; Pathak, VP; Renick, J; Soll, R; Splittgerber, U; Wrasidlo, W; Zeng, B; Zhao, N; Zhou, Y Discovery and preliminary structure-activity relationship studies of novel benzotriazine based compounds as Src inhibitors. Bioorg Med Chem Lett16:5546-50 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase Yes |
|---|
| Name: | Tyrosine-protein kinase Yes |
|---|
| Synonyms: | Proto-oncogene c-Yes | VHL/Tyrosine-protein kinase Yes | YES | YES1 | YES_HUMAN | p61-yes |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 60801.54 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P07947 |
|---|
| Residue: | 543 |
|---|
| Sequence: | MGCIKSKENKSPAIKYRPENTPEPVSTSVSHYGAEPTTVSPCPSSSAKGTAVNFSSLSMT
PFGGSSGVTPFGGASSSFSVVPSSYPAGLTGGVTIFVALYDYEARTTEDLSFKKGERFQI
INNTEGDWWEARSIATGKNGYIPSNYVAPADSIQAEEWYFGKMGRKDAERLLLNPGNQRG
IFLVRESETTKGAYSLSIRDWDEIRGDNVKHYKIRKLDNGGYYITTRAQFDTLQKLVKHY
TEHADGLCHKLTTVCPTVKPQTQGLAKDAWEIPRESLRLEVKLGQGCFGEVWMGTWNGTT
KVAIKTLKPGTMMPEAFLQEAQIMKKLRHDKLVPLYAVVSEEPIYIVTEFMSKGSLLDFL
KEGDGKYLKLPQLVDMAAQIADGMAYIERMNYIHRDLRAANILVGENLVCKIADFGLARL
IEDNEYTARQGAKFPIKWTAPEAALYGRFTIKSDVWSFGILQTELVTKGRVPYPGMVNRE
VLEQVERGYRMPCPQGCPESLHELMNLCWKKDPDERPTFEYIQSFLEDYFTATEPQYQPG
ENL
|
|
|
|---|
| BDBM50193874 |
|---|
| n/a |
|---|
| Name | BDBM50193874 |
|---|
| Synonyms: | 7-(2,6-dimethylphenyl)-5-methyl-N-(4-(2-(pyrrolidin-1-yl)ethoxy)phenyl)benzo[e][1,2,4]triazin-3-amine | 7-(2,6-dimethylphenyl)-5-methyl-N-[4-(2-pyrrolidin-1-ylethoxy)phenyl]-1,2,4-benzotriazin-3-amine | CHEMBL436137 | US8481536, 322 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H31N5O |
|---|
| Mol. Mass. | 453.5786 |
|---|
| SMILES | Cc1cccc(C)c1-c1cc(C)c2nc(Nc3ccc(OCCN4CCCC4)cc3)nnc2c1 |
|---|
| Structure | 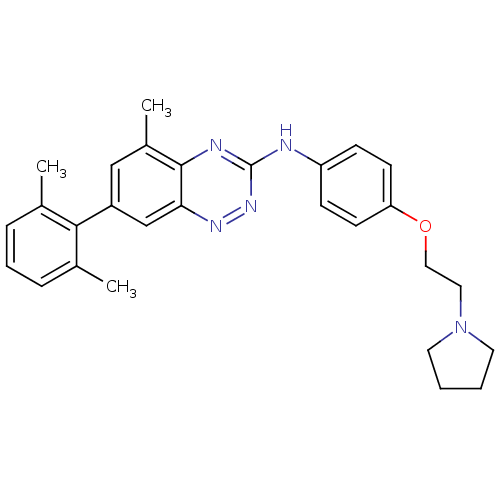
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Noronha, G; Barrett, K; Cao, J; Dneprovskaia, E; Fine, R; Gong, X; Gritzen, C; Hood, J; Kang, X; Klebansky, B; Li, G; Liao, W; Lohse, D; Mak, CC; McPherson, A; Palanki, MS; Pathak, VP; Renick, J; Soll, R; Splittgerber, U; Wrasidlo, W; Zeng, B; Zhao, N; Zhou, Y Discovery and preliminary structure-activity relationship studies of novel benzotriazine based compounds as Src inhibitors. Bioorg Med Chem Lett16:5546-50 (2006) [PubMed] Article
Noronha, G; Barrett, K; Cao, J; Dneprovskaia, E; Fine, R; Gong, X; Gritzen, C; Hood, J; Kang, X; Klebansky, B; Li, G; Liao, W; Lohse, D; Mak, CC; McPherson, A; Palanki, MS; Pathak, VP; Renick, J; Soll, R; Splittgerber, U; Wrasidlo, W; Zeng, B; Zhao, N; Zhou, Y Discovery and preliminary structure-activity relationship studies of novel benzotriazine based compounds as Src inhibitors. Bioorg Med Chem Lett16:5546-50 (2006) [PubMed] Article