| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 9 |
|---|
| Ligand | BDBM15918 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_453170 (CHEMBL902321) |
|---|
| IC50 | 74±n/a nM |
|---|
| Citation |  Liu, G; Zhao, H; Liu, B; Xin, Z; Liu, M; Serby, MD; Lubbers, NL; Widomski, DL; Polakowski, JS; Beno, DW; Trevillyan, JM; Sham, HL Hemodynamic effects of potent and selective JNK inhibitors in anesthetized rats: implication for targeting protein kinases in metabolic diseases. Bioorg Med Chem Lett17:495-500 (2007) [PubMed] Article Liu, G; Zhao, H; Liu, B; Xin, Z; Liu, M; Serby, MD; Lubbers, NL; Widomski, DL; Polakowski, JS; Beno, DW; Trevillyan, JM; Sham, HL Hemodynamic effects of potent and selective JNK inhibitors in anesthetized rats: implication for targeting protein kinases in metabolic diseases. Bioorg Med Chem Lett17:495-500 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 9 |
|---|
| Name: | Mitogen-activated protein kinase 9 |
|---|
| Synonyms: | JNK-55 | JNK2 | JNK2/JNK3 | MAPK9 | MK09_HUMAN | Mitogen-Activated Protein Kinase 9 (JNK2) | Mitogen-activated protein kinase 8/9 | PRKM9 | SAPK1A | Stress-activated protein kinase JNK2 | c-Jun N-terminal kinase 2 | c-Jun N-terminal kinase 2 (JNK2) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48131.49 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | JNK-2 was purchased from Upstate Cell Signaling Solutions (formerly Upstate Biotechnology). |
|---|
| Residue: | 424 |
|---|
| Sequence: | MSDSKCDSQFYSVQVADSTFTVLKRYQQLKPIGSGAQGIVCAAFDTVLGINVAVKKLSRP
FQNQTHAKRAYRELVLLKCVNHKNIISLLNVFTPQKTLEEFQDVYLVMELMDANLCQVIH
MELDHERMSYLLYQMLCGIKHLHSAGIIHRDLKPSNIVVKSDCTLKILDFGLARTACTNF
MMTPYVVTRYYRAPEVILGMGYKENVDIWSVGCIMGELVKGCVIFQGTDHIDQWNKVIEQ
LGTPSAEFMKKLQPTVRNYVENRPKYPGIKFEELFPDWIFPSESERDKIKTSQARDLLSK
MLVIDPDKRISVDEALRHPYITVWYDPAEAEAPPPQIYDAQLEEREHAIEEWKELIYKEV
MDWEERSKNGVVKDQPSDAAVSSNATPSQSSSINDISSMSTEQTLASDTDSSLDASTGPL
EGCR
|
|
|
|---|
| BDBM15918 |
|---|
| n/a |
|---|
| Name | BDBM15918 |
|---|
| Synonyms: | 2-pyridinecarboxamide deriv. 12 | 4-Amino-5-chloro-6-ethoxy-pyridine-2-carboxylic acid 4-methanesulfonyl-benzylamide | 4-amino-5-chloro-6-ethoxy-N-[(4-methanesulfonylphenyl)methyl]pyridine-2-carboxamide | CHEMBL377383 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H18ClN3O4S |
|---|
| Mol. Mass. | 383.85 |
|---|
| SMILES | CCOc1nc(cc(N)c1Cl)C(=O)NCc1ccc(cc1)S(C)(=O)=O |
|---|
| Structure | 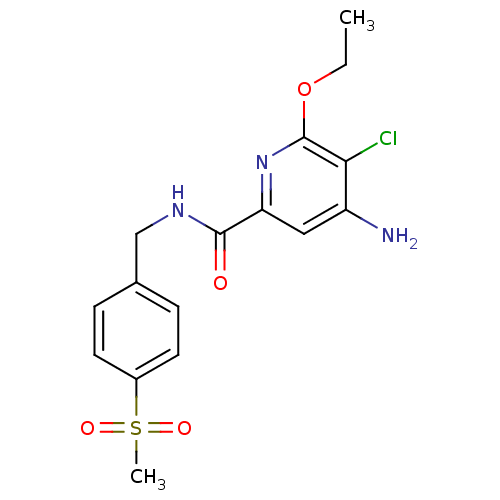
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, G; Zhao, H; Liu, B; Xin, Z; Liu, M; Serby, MD; Lubbers, NL; Widomski, DL; Polakowski, JS; Beno, DW; Trevillyan, JM; Sham, HL Hemodynamic effects of potent and selective JNK inhibitors in anesthetized rats: implication for targeting protein kinases in metabolic diseases. Bioorg Med Chem Lett17:495-500 (2007) [PubMed] Article
Liu, G; Zhao, H; Liu, B; Xin, Z; Liu, M; Serby, MD; Lubbers, NL; Widomski, DL; Polakowski, JS; Beno, DW; Trevillyan, JM; Sham, HL Hemodynamic effects of potent and selective JNK inhibitors in anesthetized rats: implication for targeting protein kinases in metabolic diseases. Bioorg Med Chem Lett17:495-500 (2007) [PubMed] Article