| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50203306 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_425259 (CHEMBL856835) |
|---|
| IC50 | >25000±n/a nM |
|---|
| Citation |  Liverton, NJ; Bednar, RA; Bednar, B; Butcher, JW; Claiborne, CF; Claremon, DA; Cunningham, M; DiLella, AG; Gaul, SL; Libby, BE; Lyle, EA; Lynch, JJ; McCauley, JA; Mosser, SD; Nguyen, KT; Stump, GL; Sun, H; Wang, H; Yergey, J; Koblan, KS Identification and characterization of 4-methylbenzyl 4-[(pyrimidin-2-ylamino)methyl]piperidine-1-carboxylate, an orally bioavailable, brain penetrant NR2B selective N-methyl-D-aspartate receptor antagonist. J Med Chem50:807-19 (2007) [PubMed] Article Liverton, NJ; Bednar, RA; Bednar, B; Butcher, JW; Claiborne, CF; Claremon, DA; Cunningham, M; DiLella, AG; Gaul, SL; Libby, BE; Lyle, EA; Lynch, JJ; McCauley, JA; Mosser, SD; Nguyen, KT; Stump, GL; Sun, H; Wang, H; Yergey, J; Koblan, KS Identification and characterization of 4-methylbenzyl 4-[(pyrimidin-2-ylamino)methyl]piperidine-1-carboxylate, an orally bioavailable, brain penetrant NR2B selective N-methyl-D-aspartate receptor antagonist. J Med Chem50:807-19 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50203306 |
|---|
| n/a |
|---|
| Name | BDBM50203306 |
|---|
| Synonyms: | CHEMBL219113 | benzyl 4-[(1,3,4-thiadiazol-2-ylamino)methyl]piperidine-1-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H20N4O2S |
|---|
| Mol. Mass. | 332.421 |
|---|
| SMILES | O=C(OCc1ccccc1)N1CCC(CNc2nncs2)CC1 |
|---|
| Structure | 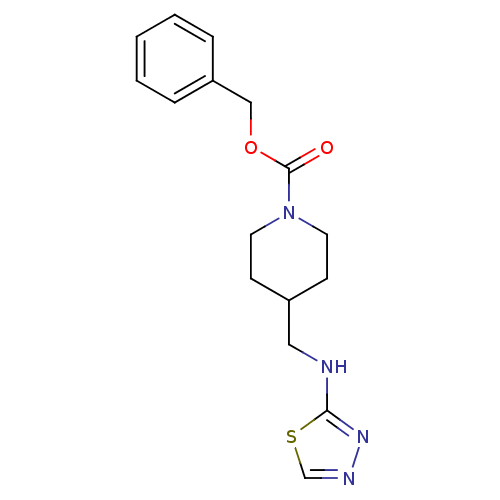
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liverton, NJ; Bednar, RA; Bednar, B; Butcher, JW; Claiborne, CF; Claremon, DA; Cunningham, M; DiLella, AG; Gaul, SL; Libby, BE; Lyle, EA; Lynch, JJ; McCauley, JA; Mosser, SD; Nguyen, KT; Stump, GL; Sun, H; Wang, H; Yergey, J; Koblan, KS Identification and characterization of 4-methylbenzyl 4-[(pyrimidin-2-ylamino)methyl]piperidine-1-carboxylate, an orally bioavailable, brain penetrant NR2B selective N-methyl-D-aspartate receptor antagonist. J Med Chem50:807-19 (2007) [PubMed] Article
Liverton, NJ; Bednar, RA; Bednar, B; Butcher, JW; Claiborne, CF; Claremon, DA; Cunningham, M; DiLella, AG; Gaul, SL; Libby, BE; Lyle, EA; Lynch, JJ; McCauley, JA; Mosser, SD; Nguyen, KT; Stump, GL; Sun, H; Wang, H; Yergey, J; Koblan, KS Identification and characterization of 4-methylbenzyl 4-[(pyrimidin-2-ylamino)methyl]piperidine-1-carboxylate, an orally bioavailable, brain penetrant NR2B selective N-methyl-D-aspartate receptor antagonist. J Med Chem50:807-19 (2007) [PubMed] Article