| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50208007 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_429176 (CHEMBL913811) |
|---|
| IC50 | 4200±n/a nM |
|---|
| Citation |  Peretto, I; Forlani, R; Fossati, C; Giardina, GA; Giardini, A; Guala, M; La Porta, E; Petrillo, P; Radaelli, S; Radice, L; Raveglia, LF; Santoro, E; Scudellaro, R; Scarpitta, F; Bigogno, C; Misiano, P; Dondio, GM; Rizzi, A; Armani, E; Amari, G; Civelli, M; Villetti, G; Patacchini, R; Bergamaschi, M; Delcanale, M; Salcedo, C; Fernández, AG; Imbimbo, BP Discovery of diaryl imidazolidin-2-one derivatives, a novel class of muscarinic M3 selective antagonists (Part 1). J Med Chem50:1571-83 (2007) [PubMed] Article Peretto, I; Forlani, R; Fossati, C; Giardina, GA; Giardini, A; Guala, M; La Porta, E; Petrillo, P; Radaelli, S; Radice, L; Raveglia, LF; Santoro, E; Scudellaro, R; Scarpitta, F; Bigogno, C; Misiano, P; Dondio, GM; Rizzi, A; Armani, E; Amari, G; Civelli, M; Villetti, G; Patacchini, R; Bergamaschi, M; Delcanale, M; Salcedo, C; Fernández, AG; Imbimbo, BP Discovery of diaryl imidazolidin-2-one derivatives, a novel class of muscarinic M3 selective antagonists (Part 1). J Med Chem50:1571-83 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50208007 |
|---|
| n/a |
|---|
| Name | BDBM50208007 |
|---|
| Synonyms: | 3-(1-(3-chlorobenzyl)-piperidin-4-yl)-5,5-di-(4-fluorophenyl)-imidazolidin-2-one | CHEMBL223079 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H26ClF2N3O |
|---|
| Mol. Mass. | 481.965 |
|---|
| SMILES | Fc1ccc(cc1)C1(CN(C2CCN(Cc3cccc(Cl)c3)CC2)C(=O)N1)c1ccc(F)cc1 |
|---|
| Structure | 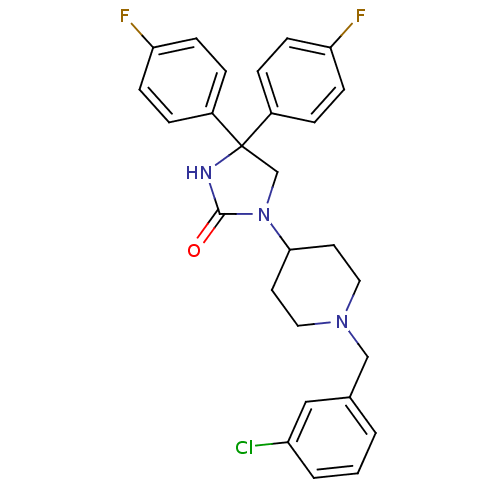
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Peretto, I; Forlani, R; Fossati, C; Giardina, GA; Giardini, A; Guala, M; La Porta, E; Petrillo, P; Radaelli, S; Radice, L; Raveglia, LF; Santoro, E; Scudellaro, R; Scarpitta, F; Bigogno, C; Misiano, P; Dondio, GM; Rizzi, A; Armani, E; Amari, G; Civelli, M; Villetti, G; Patacchini, R; Bergamaschi, M; Delcanale, M; Salcedo, C; Fernández, AG; Imbimbo, BP Discovery of diaryl imidazolidin-2-one derivatives, a novel class of muscarinic M3 selective antagonists (Part 1). J Med Chem50:1571-83 (2007) [PubMed] Article
Peretto, I; Forlani, R; Fossati, C; Giardina, GA; Giardini, A; Guala, M; La Porta, E; Petrillo, P; Radaelli, S; Radice, L; Raveglia, LF; Santoro, E; Scudellaro, R; Scarpitta, F; Bigogno, C; Misiano, P; Dondio, GM; Rizzi, A; Armani, E; Amari, G; Civelli, M; Villetti, G; Patacchini, R; Bergamaschi, M; Delcanale, M; Salcedo, C; Fernández, AG; Imbimbo, BP Discovery of diaryl imidazolidin-2-one derivatives, a novel class of muscarinic M3 selective antagonists (Part 1). J Med Chem50:1571-83 (2007) [PubMed] Article