| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50219241 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_437417 (CHEMBL906815) |
|---|
| IC50 | 2450±n/a nM |
|---|
| Citation |  Kwon, YE; Park, JY; No, KT; Shin, JH; Lee, SK; Eun, JS; Yang, JH; Shin, TY; Kim, DK; Chae, BS; Leem, JY; Kim, KH Synthesis, in vitro assay, and molecular modeling of new piperidine derivatives having dual inhibitory potency against acetylcholinesterase and Abeta1-42 aggregation for Alzheimer's disease therapeutics. Bioorg Med Chem15:6596-607 (2007) [PubMed] Article Kwon, YE; Park, JY; No, KT; Shin, JH; Lee, SK; Eun, JS; Yang, JH; Shin, TY; Kim, DK; Chae, BS; Leem, JY; Kim, KH Synthesis, in vitro assay, and molecular modeling of new piperidine derivatives having dual inhibitory potency against acetylcholinesterase and Abeta1-42 aggregation for Alzheimer's disease therapeutics. Bioorg Med Chem15:6596-607 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_ELEEL | Acetylcholinesterase (AChE) | Acetylcholinesterase (EeAChE) | ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 71812.79 |
|---|
| Organism: | Electrophorus electricus (Electric eel) |
|---|
| Description: | n/a |
|---|
| Residue: | 633 |
|---|
| Sequence: | MKILDALLFPVIFIMFFIHLSIAQTDPELTIMTRLGQVQGTRLPVPDRSHVIAFLGIPFA
EPPLGKMRFKPPEPKKPWNDVFDARDYPSACYQYVDTSYPGFSGTEMWNPNRMMSEDCLY
LNVWVPATPRPHNLTVMVWIYGGGFYSGSSSLDVYDGRYLAHSEKVVVVSMNYRVSAFGF
LALNGSAEAPGNVGLLDQRLALQWVQDNIHFFGGNPKQVTIFGESAGAASVGMHLLSPDS
RPKFTRAILQSGVPNGPWRTVSFDEARRRAIKLGRLVGCPDGNDTDLIDCLRSKQPQDLI
DQEWLVLPFSGLFRFSFVPVIDGVVFPDTPEAMLNSGNFKDTQILLGVNQNEGSYFLIYG
APGFSKDNESLITREDFLQGVKMSVPHANEIGLEAVILQYTDWMDEDNPIKNREAMDDIV
GDHNVVCPLQHFAKMYAQYSILQGQTGTASQGNLGWGNSGSASNSGNSQVSVYLYMFDHR
ASNLVWPEWMGVIHGYEIEFVFGLPLEKRLNYTLEEEKLSRRMMKYWANFARTGNPNINV
DGSIDSRRRWPVFTSTEQKHVGLNTDSLKVHKGLKSQFCALWNRFLPRLLNVTENIDDAE
RQWKAEFHRWSSYMMHWKNQFDHYSKQERCTNL
|
|
|
|---|
| BDBM50219241 |
|---|
| n/a |
|---|
| Name | BDBM50219241 |
|---|
| Synonyms: | 4-[4-(benzhydryloxy)piperidino]butyl-N-(4-fluorophenyl)carbamate | CHEMBL426915 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H33FN2O3 |
|---|
| Mol. Mass. | 476.5823 |
|---|
| SMILES | Fc1ccc(NC(=O)OCCCCN2CCC(CC2)OC(c2ccccc2)c2ccccc2)cc1 |
|---|
| Structure | 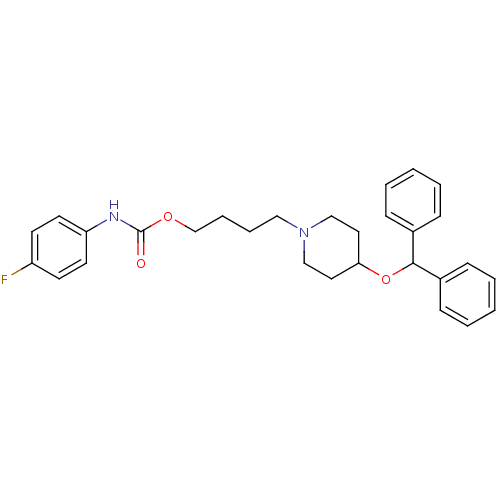
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kwon, YE; Park, JY; No, KT; Shin, JH; Lee, SK; Eun, JS; Yang, JH; Shin, TY; Kim, DK; Chae, BS; Leem, JY; Kim, KH Synthesis, in vitro assay, and molecular modeling of new piperidine derivatives having dual inhibitory potency against acetylcholinesterase and Abeta1-42 aggregation for Alzheimer's disease therapeutics. Bioorg Med Chem15:6596-607 (2007) [PubMed] Article
Kwon, YE; Park, JY; No, KT; Shin, JH; Lee, SK; Eun, JS; Yang, JH; Shin, TY; Kim, DK; Chae, BS; Leem, JY; Kim, KH Synthesis, in vitro assay, and molecular modeling of new piperidine derivatives having dual inhibitory potency against acetylcholinesterase and Abeta1-42 aggregation for Alzheimer's disease therapeutics. Bioorg Med Chem15:6596-607 (2007) [PubMed] Article