| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proteinase-activated receptor 1 |
|---|
| Ligand | BDBM50222029 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_446321 (CHEMBL895431) |
|---|
| IC50 | 224±n/a nM |
|---|
| Citation |  Chelliah, MV; Chackalamannil, S; Xia, Y; Eagen, K; Clasby, MC; Gao, X; Greenlee, W; Ahn, HS; Agans-Fantuzzi, J; Boykow, G; Hsieh, Y; Bryant, M; Palamanda, J; Chan, TM; Hesk, D; Chintala, M Heterotricyclic himbacine analogs as potent, orally active thrombin receptor (protease activated receptor-1) antagonists. J Med Chem50:5147-60 (2007) [PubMed] Article Chelliah, MV; Chackalamannil, S; Xia, Y; Eagen, K; Clasby, MC; Gao, X; Greenlee, W; Ahn, HS; Agans-Fantuzzi, J; Boykow, G; Hsieh, Y; Bryant, M; Palamanda, J; Chan, TM; Hesk, D; Chintala, M Heterotricyclic himbacine analogs as potent, orally active thrombin receptor (protease activated receptor-1) antagonists. J Med Chem50:5147-60 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proteinase-activated receptor 1 |
|---|
| Name: | Proteinase-activated receptor 1 |
|---|
| Synonyms: | CF2R | Coagulation factor II receptor | F2R | PAR-1 | PAR1 | PAR1_HUMAN | Proteinase activated receptor 1 | Proteinase-activated receptor 1 (PAR-1) | TR | Thrombin receptor | Thrombin receptor/ Proteinase-activated receptor 1(Par-1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 47450.07 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P25116 |
|---|
| Residue: | 425 |
|---|
| Sequence: | MGPRRLLLVAACFSLCGPLLSARTRARRPESKATNATLDPRSFLLRNPNDKYEPFWEDEE
KNESGLTEYRLVSINKSSPLQKQLPAFISEDASGYLTSSWLTLFVPSVYTGVFVVSLPLN
IMAIVVFILKMKVKKPAVVYMLHLATADVLFVSVLPFKISYYFSGSDWQFGSELCRFVTA
AFYCNMYASILLMTVISIDRFLAVVYPMQSLSWRTLGRASFTCLAIWALAIAGVVPLLLK
EQTIQVPGLNITTCHDVLNETLLEGYYAYYFSAFSAVFFFVPLIISTVCYVSIIRCLSSS
AVANRSKKSRALFLSAAVFCIFIICFGPTNVLLIAHYSFLSHTSTTEAAYFAYLLCVCVS
SISCCIDPLIYYYASSECQRYVYSILCCKESSDPSSYNSSGQLMASKMDTCSSNLNNSIY
KKLLT
|
|
|
|---|
| BDBM50222029 |
|---|
| n/a |
|---|
| Name | BDBM50222029 |
|---|
| Synonyms: | CHEMBL244484 | ethyl (4aS,5S,5aS,6R,8aR,9aS)-5-[(E)-2-[5-(3-fluorophenyl)-2-pyridinyl]ethenyl]-decahydro-6-methyl-8-oxofuro[3,4-g]quinoline-1(2H)-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H31FN2O4 |
|---|
| Mol. Mass. | 478.5551 |
|---|
| SMILES | CCOC(=O)N1CCC[C@@H]2[C@@H]1C[C@@H]1[C@@H]([C@@H](C)OC1=O)[C@H]2\C=C\c1ccc(cn1)-c1cccc(F)c1 |
|---|
| Structure | 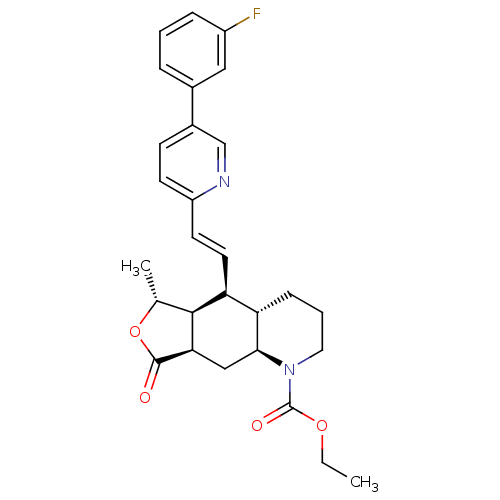
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chelliah, MV; Chackalamannil, S; Xia, Y; Eagen, K; Clasby, MC; Gao, X; Greenlee, W; Ahn, HS; Agans-Fantuzzi, J; Boykow, G; Hsieh, Y; Bryant, M; Palamanda, J; Chan, TM; Hesk, D; Chintala, M Heterotricyclic himbacine analogs as potent, orally active thrombin receptor (protease activated receptor-1) antagonists. J Med Chem50:5147-60 (2007) [PubMed] Article
Chelliah, MV; Chackalamannil, S; Xia, Y; Eagen, K; Clasby, MC; Gao, X; Greenlee, W; Ahn, HS; Agans-Fantuzzi, J; Boykow, G; Hsieh, Y; Bryant, M; Palamanda, J; Chan, TM; Hesk, D; Chintala, M Heterotricyclic himbacine analogs as potent, orally active thrombin receptor (protease activated receptor-1) antagonists. J Med Chem50:5147-60 (2007) [PubMed] Article