| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 72 kDa type IV collagenase |
|---|
| Ligand | BDBM11551 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_461906 (CHEMBL944753) |
|---|
| Ki | 0.400000±n/a nM |
|---|
| Citation |  Yang, SM; Scannevin, RH; Wang, B; Burke, SL; Huang, Z; Karnachi, P; Wilson, LJ; Rhodes, KJ; Lagu, B; Murray, WV beta-N-Biaryl ether sulfonamide hydroxamates as potent gelatinase inhibitors: part 2. Optimization of alpha-amino substituents. Bioorg Med Chem Lett18:1140-5 (2008) [PubMed] Article Yang, SM; Scannevin, RH; Wang, B; Burke, SL; Huang, Z; Karnachi, P; Wilson, LJ; Rhodes, KJ; Lagu, B; Murray, WV beta-N-Biaryl ether sulfonamide hydroxamates as potent gelatinase inhibitors: part 2. Optimization of alpha-amino substituents. Bioorg Med Chem Lett18:1140-5 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 72 kDa type IV collagenase |
|---|
| Name: | 72 kDa type IV collagenase |
|---|
| Synonyms: | 72 kDa gelatinase | 72 kDa type IV collagenase precursor | CLG4A | Gelatinase A | Gelatinase A (MMP-2) | MMP2 | MMP2_HUMAN | Matrix metalloproteinase-2 | Matrix metalloproteinase-2 (MMP 2) | Matrix metalloproteinase-2 (MMP2) | Matrix metalloproteinases 2 (MMP-2) | TBE-1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 73870.36 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08253 |
|---|
| Residue: | 660 |
|---|
| Sequence: | MEALMARGALTGPLRALCLLGCLLSHAAAAPSPIIKFPGDVAPKTDKELAVQYLNTFYGC
PKESCNLFVLKDTLKKMQKFFGLPQTGDLDQNTIETMRKPRCGNPDVANYNFFPRKPKWD
KNQITYRIIGYTPDLDPETVDDAFARAFQVWSDVTPLRFSRIHDGEADIMINFGRWEHGD
GYPFDGKDGLLAHAFAPGTGVGGDSHFDDDELWTLGEGQVVRVKYGNADGEYCKFPFLFN
GKEYNSCTDTGRSDGFLWCSTTYNFEKDGKYGFCPHEALFTMGGNAEGQPCKFPFRFQGT
SYDSCTTEGRTDGYRWCGTTEDYDRDKKYGFCPETAMSTVGGNSEGAPCVFPFTFLGNKY
ESCTSAGRSDGKMWCATTANYDDDRKWGFCPDQGYSLFLVAAHEFGHAMGLEHSQDPGAL
MAPIYTYTKNFRLSQDDIKGIQELYGASPDIDLGTGPTPTLGPVTPEICKQDIVFDGIAQ
IRGEIFFFKDRFIWRTVTPRDKPMGPLLVATFWPELPEKIDAVYEAPQEEKAVFFAGNEY
WIYSASTLERGYPKPLTSLGLPPDVQRVDAAFNWSKNKKTYIFAGDKFWRYNEVKKKMDP
GFPKLIADAWNAIPDNLDAVVDLQGGGHSYFFKGAYYLKLENQSLKSVKFGSIKSDWLGC
|
|
|
|---|
| BDBM11551 |
|---|
| n/a |
|---|
| Name | BDBM11551 |
|---|
| Synonyms: | (3R)-N-hydroxy-2-[(4-methoxy-1,1-biphenyl-4-yl)methyl]-1,2-thiazinane-3-carboxamide 1,1-dioxide | (3R)-N-hydroxy-2-{[4-(4-methoxyphenyl)phenyl]methyl}-1,1-dioxo-1,2-thiazinane-3-carboxamide | CHEMBL103418 | Sultam Hydroxamate 23a |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H22N2O5S |
|---|
| Mol. Mass. | 390.453 |
|---|
| SMILES | COc1ccc(cc1)-c1ccc(CN2[C@H](CCCS2(=O)=O)C(=O)NO)cc1 |r| |
|---|
| Structure | 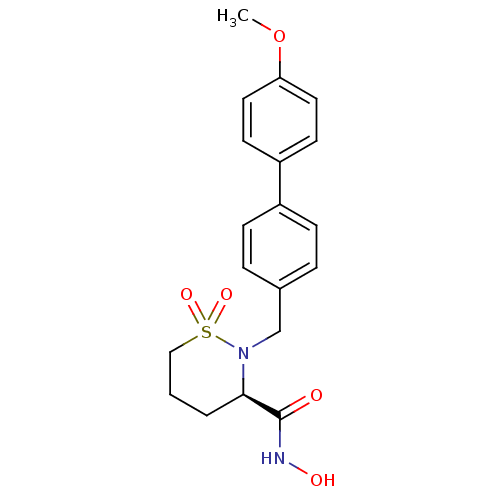
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yang, SM; Scannevin, RH; Wang, B; Burke, SL; Huang, Z; Karnachi, P; Wilson, LJ; Rhodes, KJ; Lagu, B; Murray, WV beta-N-Biaryl ether sulfonamide hydroxamates as potent gelatinase inhibitors: part 2. Optimization of alpha-amino substituents. Bioorg Med Chem Lett18:1140-5 (2008) [PubMed] Article
Yang, SM; Scannevin, RH; Wang, B; Burke, SL; Huang, Z; Karnachi, P; Wilson, LJ; Rhodes, KJ; Lagu, B; Murray, WV beta-N-Biaryl ether sulfonamide hydroxamates as potent gelatinase inhibitors: part 2. Optimization of alpha-amino substituents. Bioorg Med Chem Lett18:1140-5 (2008) [PubMed] Article