| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D-aspartate oxidase |
|---|
| Ligand | BDBM50004955 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_556667 (CHEMBL958954) |
|---|
| IC50 | >5000±n/a nM |
|---|
| Citation |  Sparey, T; Abeywickrema, P; Almond, S; Brandon, N; Byrne, N; Campbell, A; Hutson, PH; Jacobson, M; Jones, B; Munshi, S; Pascarella, D; Pike, A; Prasad, GS; Sachs, N; Sakatis, M; Sardana, V; Venkatraman, S; Young, MB The discovery of fused pyrrole carboxylic acids as novel, potent D-amino acid oxidase (DAO) inhibitors. Bioorg Med Chem Lett18:3386-91 (2008) [PubMed] Article Sparey, T; Abeywickrema, P; Almond, S; Brandon, N; Byrne, N; Campbell, A; Hutson, PH; Jacobson, M; Jones, B; Munshi, S; Pascarella, D; Pike, A; Prasad, GS; Sachs, N; Sakatis, M; Sardana, V; Venkatraman, S; Young, MB The discovery of fused pyrrole carboxylic acids as novel, potent D-amino acid oxidase (DAO) inhibitors. Bioorg Med Chem Lett18:3386-91 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D-aspartate oxidase |
|---|
| Name: | D-aspartate oxidase |
|---|
| Synonyms: | D-Aspartate Oxidase (DDO) | DASOX | DDO | OXDD_HUMAN |
|---|
| Type: | Oxidoreductase |
|---|
| Mol. Mass.: | 37543.06 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant human DDO expressed and purified from sf9 insect cells, was used in enzyme assays. |
|---|
| Residue: | 341 |
|---|
| Sequence: | MDTARIAVVGAGVVGLSTAVCISKLVPRCSVTIISDKFTPDTTSDVAAGMLIPHTYPDTP
IHTQKQWFRETFNHLFAIANSAEAGDAGVHLVSGWQIFQSTPTEEVPFWADVVLGFRKMT
EAELKKFPQYVFGQAFTTLKCECPAYLPWLEKRIKGSGGWTLTRRIEDLWELHPSFDIVV
NCSGLGSRQLAGDSKIFPVRGQVLQVQAPWVEHFIRDGSGLTYIYPGTSHVTLGGTRQKG
DWNLSPDAENSREILSRCCALEPSLHGACNIREKVGLRPYRPGVRLQTELLARDGQRLPV
VHHYGHGSGGISVHWGTALEAARLVSECVHALRTPIPKSNL
|
|
|
|---|
| BDBM50004955 |
|---|
| n/a |
|---|
| Name | BDBM50004955 |
|---|
| Synonyms: | 1H-Indole-2-carboxylic acid | CHEMBL278390 | Indol-2-carboxylic acid | Indole-2-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H7NO2 |
|---|
| Mol. Mass. | 161.1574 |
|---|
| SMILES | OC(=O)c1cc2ccccc2[nH]1 |
|---|
| Structure | 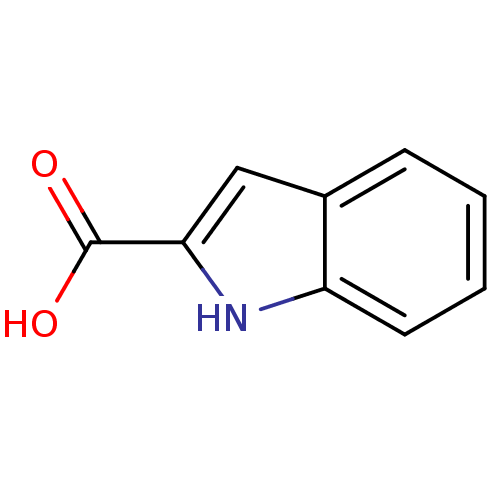
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sparey, T; Abeywickrema, P; Almond, S; Brandon, N; Byrne, N; Campbell, A; Hutson, PH; Jacobson, M; Jones, B; Munshi, S; Pascarella, D; Pike, A; Prasad, GS; Sachs, N; Sakatis, M; Sardana, V; Venkatraman, S; Young, MB The discovery of fused pyrrole carboxylic acids as novel, potent D-amino acid oxidase (DAO) inhibitors. Bioorg Med Chem Lett18:3386-91 (2008) [PubMed] Article
Sparey, T; Abeywickrema, P; Almond, S; Brandon, N; Byrne, N; Campbell, A; Hutson, PH; Jacobson, M; Jones, B; Munshi, S; Pascarella, D; Pike, A; Prasad, GS; Sachs, N; Sakatis, M; Sardana, V; Venkatraman, S; Young, MB The discovery of fused pyrrole carboxylic acids as novel, potent D-amino acid oxidase (DAO) inhibitors. Bioorg Med Chem Lett18:3386-91 (2008) [PubMed] Article