

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Transcription factor Jun | ||
| Ligand | BDBM50279698 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_545230 (CHEMBL1019824) | ||
| IC50 | 5500±n/a nM | ||
| Citation |  Giri, RS; Thaker, HM; Giordano, T; Williams, J; Rogers, D; Sudersanam, V; Vasu, KK Design, synthesis and characterization of novel 2-(2,4-disubstituted-thiazole-5-yl)-3-aryl-3H-quinazoline-4-one derivatives as inhibitors of NF-kappaB and AP-1 mediated transcription activation and as potential anti-inflammatory agents. Eur J Med Chem44:2184-9 (2009) [PubMed] Article Giri, RS; Thaker, HM; Giordano, T; Williams, J; Rogers, D; Sudersanam, V; Vasu, KK Design, synthesis and characterization of novel 2-(2,4-disubstituted-thiazole-5-yl)-3-aryl-3H-quinazoline-4-one derivatives as inhibitors of NF-kappaB and AP-1 mediated transcription activation and as potential anti-inflammatory agents. Eur J Med Chem44:2184-9 (2009) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Transcription factor Jun | |||
| Name: | Transcription factor Jun | ||
| Synonyms: | AP1 | Activator protein 1 | JUN | JUN_HUMAN | Proto-oncogene c-JUN | Transcription factor AP-1 | Transcription factor AP1 | V-jun avian sarcoma virus 17 oncogene homolog | p39 | ||
| Type: | n/a | ||
| Mol. Mass.: | 35683.24 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P05412 | ||
| Residue: | 331 | ||
| Sequence: |
| ||
| BDBM50279698 | |||
| n/a | |||
| Name | BDBM50279698 | ||
| Synonyms: | 2-(4-Methyl-2-methylamino-thiazol-5-yl)-3-o-tolyl-3H-quinazolin-4-one | CHEMBL515276 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C20H18N4OS | ||
| Mol. Mass. | 362.448 | ||
| SMILES | CNc1nc(C)c(s1)-c1nc2ccccc2c(=O)n1-c1ccccc1C |(32.14,-.5,;31.37,.83,;29.84,.83,;29.07,-.51,;27.56,-.19,;26.42,-1.22,;27.4,1.34,;28.8,1.97,;26.07,2.11,;24.72,1.33,;23.38,2.1,;22.04,1.33,;20.71,2.1,;20.71,3.65,;22.04,4.42,;23.38,3.65,;24.71,4.44,;24.7,5.98,;26.06,3.66,;27.39,4.44,;28.73,3.67,;30.06,4.44,;30.05,5.98,;28.71,6.75,;27.38,5.97,;26.05,6.73,)| | ||
| Structure | 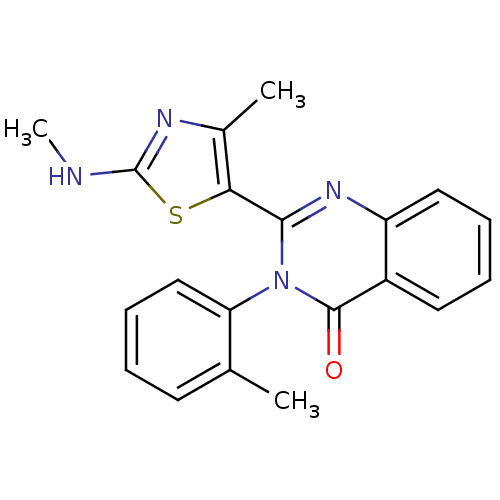
| ||