| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Epidermal growth factor receptor |
|---|
| Ligand | BDBM50277623 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_501537 (CHEMBL984177) |
|---|
| IC50 | 5400±n/a nM |
|---|
| Citation |  Cirillo, PF; Hickey, ER; Moss, N; Breitfelder, S; Betageri, R; Fadra, T; Gaenzler, F; Gilmore, T; Goldberg, DR; Kamhi, V; Kirrane, T; Kroe, RR; Madwed, J; Moriak, M; Netherton, M; Pargellis, CA; Patel, UR; Qian, KC; Sharma, R; Sun, S; Swinamer, A; Torcellini, C; Takahashi, H; Tsang, M; Xiong, Z Discovery and characterization of the N-phenyl-N'-naphthylurea class of p38 kinase inhibitors. Bioorg Med Chem Lett19:2386-91 (2009) [PubMed] Article Cirillo, PF; Hickey, ER; Moss, N; Breitfelder, S; Betageri, R; Fadra, T; Gaenzler, F; Gilmore, T; Goldberg, DR; Kamhi, V; Kirrane, T; Kroe, RR; Madwed, J; Moriak, M; Netherton, M; Pargellis, CA; Patel, UR; Qian, KC; Sharma, R; Sun, S; Swinamer, A; Torcellini, C; Takahashi, H; Tsang, M; Xiong, Z Discovery and characterization of the N-phenyl-N'-naphthylurea class of p38 kinase inhibitors. Bioorg Med Chem Lett19:2386-91 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Epidermal growth factor receptor |
|---|
| Name: | Epidermal growth factor receptor |
|---|
| Synonyms: | EGF Receptor Substrate | EGFR | EGFR_HUMAN | ERBB | ERBB1 | Epidermal Growth Factor Receptor Tyrosine Kinase | Epidermal Growth Factor Receptor erbB-1 | Epidermal Growth Factor Receptor, ErbB-1 | Epidermal growth factor receptor (EGFR) | Epidermal growth factor receptor (HER1) | HER1 | Protein-Tyrosine Kinase Erbb-1 | Proto-oncogene c-ErbB-1 | Receptor tyrosine-protein kinase ErbB-1 | Tyrosine-Protein Kinase ErbB-1 |
|---|
| Type: | Receptor Kinase Domain |
|---|
| Mol. Mass.: | 134279.59 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00533 |
|---|
| Residue: | 1210 |
|---|
| Sequence: | MRPSGTAGAALLALLAALCPASRALEEKKVCQGTSNKLTQLGTFEDHFLSLQRMFNNCEV
VLGNLEITYVQRNYDLSFLKTIQEVAGYVLIALNTVERIPLENLQIIRGNMYYENSYALA
VLSNYDANKTGLKELPMRNLQEILHGAVRFSNNPALCNVESIQWRDIVSSDFLSNMSMDF
QNHLGSCQKCDPSCPNGSCWGAGEENCQKLTKIICAQQCSGRCRGKSPSDCCHNQCAAGC
TGPRESDCLVCRKFRDEATCKDTCPPLMLYNPTTYQMDVNPEGKYSFGATCVKKCPRNYV
VTDHGSCVRACGADSYEMEEDGVRKCKKCEGPCRKVCNGIGIGEFKDSLSINATNIKHFK
NCTSISGDLHILPVAFRGDSFTHTPPLDPQELDILKTVKEITGFLLIQAWPENRTDLHAF
ENLEIIRGRTKQHGQFSLAVVSLNITSLGLRSLKEISDGDVIISGNKNLCYANTINWKKL
FGTSGQKTKIISNRGENSCKATGQVCHALCSPEGCWGPEPRDCVSCRNVSRGRECVDKCN
LLEGEPREFVENSECIQCHPECLPQAMNITCTGRGPDNCIQCAHYIDGPHCVKTCPAGVM
GENNTLVWKYADAGHVCHLCHPNCTYGCTGPGLEGCPTNGPKIPSIATGMVGALLLLLVV
ALGIGLFMRRRHIVRKRTLRRLLQERELVEPLTPSGEAPNQALLRILKETEFKKIKVLGS
GAFGTVYKGLWIPEGEKVKIPVAIKELREATSPKANKEILDEAYVMASVDNPHVCRLLGI
CLTSTVQLITQLMPFGCLLDYVREHKDNIGSQYLLNWCVQIAKGMNYLEDRRLVHRDLAA
RNVLVKTPQHVKITDFGLAKLLGAEEKEYHAEGGKVPIKWMALESILHRIYTHQSDVWSY
GVTVWELMTFGSKPYDGIPASEISSILEKGERLPQPPICTIDVYMIMVKCWMIDADSRPK
FRELIIEFSKMARDPQRYLVIQGDERMHLPSPTDSNFYRALMDEEDMDDVVDADEYLIPQ
QGFFSSPSTSRTPLLSSLSATSNNSTVACIDRNGLQSCPIKEDSFLQRYSSDPTGALTED
SIDDTFLPVPEYINQSVPKRPAGSVQNPVYHNQPLNPAPSRDPHYQDPHSTAVGNPEYLN
TVQPTCVNSTFDSPAHWAQKGSHQISLDNPDYQQDFFPKEAKPNGIFKGSTAENAEYLRV
APQSSEFIGA
|
|
|
|---|
| BDBM50277623 |
|---|
| n/a |
|---|
| Name | BDBM50277623 |
|---|
| Synonyms: | CHEMBL451523 | N-(5-tert-butyl-2-methoxy-3-(3-(4-(6-(morpholinomethyl)pyridin-3-yl)naphthalen-1-yl)ureido)phenyl)methanesulfonamide | N-{5-tert-butyl-2-methoxy-3-[({4-[6-(morpholin-4-ylmethyl)pyridin-3-yl]naphthalen-1-yl}carbamoyl)amino]phenyl}methanesulfonamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H39N5O5S |
|---|
| Mol. Mass. | 617.758 |
|---|
| SMILES | COc1c(NC(=O)Nc2ccc(-c3ccc(CN4CCOCC4)nc3)c3ccccc23)cc(cc1NS(C)(=O)=O)C(C)(C)C |
|---|
| Structure | 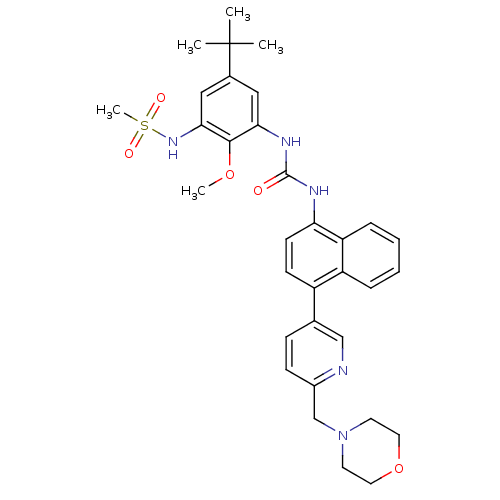
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cirillo, PF; Hickey, ER; Moss, N; Breitfelder, S; Betageri, R; Fadra, T; Gaenzler, F; Gilmore, T; Goldberg, DR; Kamhi, V; Kirrane, T; Kroe, RR; Madwed, J; Moriak, M; Netherton, M; Pargellis, CA; Patel, UR; Qian, KC; Sharma, R; Sun, S; Swinamer, A; Torcellini, C; Takahashi, H; Tsang, M; Xiong, Z Discovery and characterization of the N-phenyl-N'-naphthylurea class of p38 kinase inhibitors. Bioorg Med Chem Lett19:2386-91 (2009) [PubMed] Article
Cirillo, PF; Hickey, ER; Moss, N; Breitfelder, S; Betageri, R; Fadra, T; Gaenzler, F; Gilmore, T; Goldberg, DR; Kamhi, V; Kirrane, T; Kroe, RR; Madwed, J; Moriak, M; Netherton, M; Pargellis, CA; Patel, UR; Qian, KC; Sharma, R; Sun, S; Swinamer, A; Torcellini, C; Takahashi, H; Tsang, M; Xiong, Z Discovery and characterization of the N-phenyl-N'-naphthylurea class of p38 kinase inhibitors. Bioorg Med Chem Lett19:2386-91 (2009) [PubMed] Article