| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50025346 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_522831 (CHEMBL1002499) |
|---|
| IC50 | <400±n/a nM |
|---|
| Citation |  Lin, LS; Lanza, T; Jewell, JP; Liu, P; Jones, C; Kieczykowski, GR; Treonze, K; Si, Q; Manior, S; Koo, G; Tong, X; Wang, J; Schuelke, A; Pivnichny, J; Wang, R; Raab, C; Vincent, S; Davies, P; Maccoss, M; Mumford, RA; Hagmann, WK Discovery of N-{N-[(3-cyanophenyl)sulfonyl]-4(R)-cyclobutylamino-(L)-prolyl}-4-[(3',5'-dichloroisonicotinoyl) amino]-(L)-phenylalanine (MK-0668), an extremely potent and orally active antagonist of very late antigen-4. J Med Chem52:3449-52 (2009) [PubMed] Article Lin, LS; Lanza, T; Jewell, JP; Liu, P; Jones, C; Kieczykowski, GR; Treonze, K; Si, Q; Manior, S; Koo, G; Tong, X; Wang, J; Schuelke, A; Pivnichny, J; Wang, R; Raab, C; Vincent, S; Davies, P; Maccoss, M; Mumford, RA; Hagmann, WK Discovery of N-{N-[(3-cyanophenyl)sulfonyl]-4(R)-cyclobutylamino-(L)-prolyl}-4-[(3',5'-dichloroisonicotinoyl) amino]-(L)-phenylalanine (MK-0668), an extremely potent and orally active antagonist of very late antigen-4. J Med Chem52:3449-52 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50025346 |
|---|
| n/a |
|---|
| Name | BDBM50025346 |
|---|
| Synonyms: | CHEMBL502381 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H33Cl4N5O6S |
|---|
| Mol. Mass. | 757.511 |
|---|
| SMILES | CCOC(=O)[C@H](Cc1ccc(NC(=O)c2c(Cl)cncc2Cl)cc1)NC(=O)[C@@H]1C[C@H](CN1S(=O)(=O)c1cc(Cl)cc(Cl)c1)NC1CCC1 |r| |
|---|
| Structure | 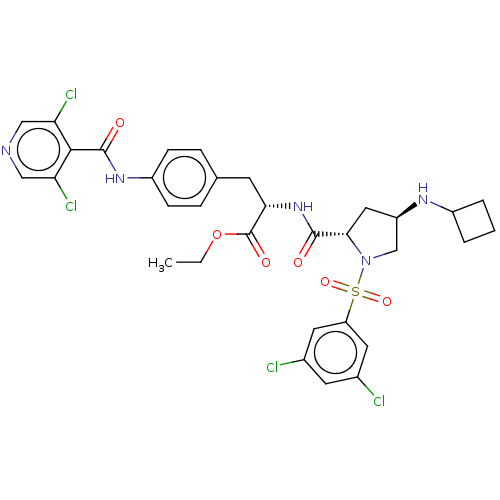
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lin, LS; Lanza, T; Jewell, JP; Liu, P; Jones, C; Kieczykowski, GR; Treonze, K; Si, Q; Manior, S; Koo, G; Tong, X; Wang, J; Schuelke, A; Pivnichny, J; Wang, R; Raab, C; Vincent, S; Davies, P; Maccoss, M; Mumford, RA; Hagmann, WK Discovery of N-{N-[(3-cyanophenyl)sulfonyl]-4(R)-cyclobutylamino-(L)-prolyl}-4-[(3',5'-dichloroisonicotinoyl) amino]-(L)-phenylalanine (MK-0668), an extremely potent and orally active antagonist of very late antigen-4. J Med Chem52:3449-52 (2009) [PubMed] Article
Lin, LS; Lanza, T; Jewell, JP; Liu, P; Jones, C; Kieczykowski, GR; Treonze, K; Si, Q; Manior, S; Koo, G; Tong, X; Wang, J; Schuelke, A; Pivnichny, J; Wang, R; Raab, C; Vincent, S; Davies, P; Maccoss, M; Mumford, RA; Hagmann, WK Discovery of N-{N-[(3-cyanophenyl)sulfonyl]-4(R)-cyclobutylamino-(L)-prolyl}-4-[(3',5'-dichloroisonicotinoyl) amino]-(L)-phenylalanine (MK-0668), an extremely potent and orally active antagonist of very late antigen-4. J Med Chem52:3449-52 (2009) [PubMed] Article