| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Ligand | BDBM50284680 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_3964 |
|---|
| IC50 | 1900±n/a nM |
|---|
| Citation |  Andrews, EG; Antognoli, GW; Breslow, R; Carta, MP; Carty, TJ; Chambers, RJ; Cheng, JB; Cohan, VL; Collins, JL; Damon, DB; Delehunt, J; Eggler, JF; Eskra, JD; Freiert, KW; Hada, WA; Marfat, A; Masamune, H; Melvin, LS; Mularski, CJ; Naclerio, BA Synthesis and pharmacological profile of two novel heterocyclic chromanols, CP-80,798 and CP-85,958, as potent LTD4 receptor antagonists Bioorg Med Chem Lett5:1365-1370 (1995) Article Andrews, EG; Antognoli, GW; Breslow, R; Carta, MP; Carty, TJ; Chambers, RJ; Cheng, JB; Cohan, VL; Collins, JL; Damon, DB; Delehunt, J; Eggler, JF; Eskra, JD; Freiert, KW; Hada, WA; Marfat, A; Masamune, H; Melvin, LS; Mularski, CJ; Naclerio, BA Synthesis and pharmacological profile of two novel heterocyclic chromanols, CP-80,798 and CP-85,958, as potent LTD4 receptor antagonists Bioorg Med Chem Lett5:1365-1370 (1995) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Name: | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Synonyms: | Alox5 | Arachidonate 5-lipoxygenase | LOX5_RAT |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 78082.31 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1432947 |
|---|
| Residue: | 673 |
|---|
| Sequence: | MPSYTVTVATGSQWFAGTDDYIYLSLIGSAGCSEKHLLDKAFYNDFERGGRDSYDVTVDE
ELGEIYLVKIEKRKYRLHDDWYLKYITLKTPHDYIEFPCYRWITGEGEIVLRDGCAKLAR
DDQIHILKQHRRKELETRQKQYRWMEWNPGFPLSIDAKCHKDLPRDIQFDSEKGVDFVLN
YSKAMENLFINRFMHMFQSSWHDFADFEKIFVKISNTISERVKNHWQEDLMFGYQFLNGC
NPVLIKRCTELPKKLPVTTEMVECSLERQLSLEQEVQEGNIFIVDYELLDGIDANKTDPC
THQFLAAPICLLYKNLANKIVPIAIQLNQTPGEKNPIFLPTDSKYDWLLAKIWVRSSDFH
IHQTITHLLRTHLVSEVFGIAMYRQLPAVHPLFKLLVAHVRFTIAINTKAREQLNCEYGL
FDKANATGGGGHVQMVQRAVQDLTYSSLCFPEAIKARGMDNTEDIPYYFYRDDGLLVWEA
IQSFTTEVVSIYYEDDQVVEEDQELQDFVKDVYVYGMRGRKASGFPKSIKSREKLSEYLT
VVIFTASAQHAAVNFGQYDWCSWIPNAPPTMRAPPPTAKGVVTIEQIVDTLPDRGRSCWH
LGAVWALSQFQENELFLGMYPEEHFIEKPVKEAMIRFRKNLEAIVSVIAERNKNKKLPYY
YLSPDRIPNSVAI
|
|
|
|---|
| BDBM50284680 |
|---|
| n/a |
|---|
| Name | BDBM50284680 |
|---|
| Synonyms: | (3S,4S)-3-Pyridin-3-ylmethyl-6-(quinolin-2-ylmethoxy)-chroman-4-ol | 3-Pyridin-3-ylmethyl-6-(quinolin-2-ylmethoxy)-chroman-4-ol | CHEMBL282524 | CP-80798 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H22N2O3 |
|---|
| Mol. Mass. | 398.4538 |
|---|
| SMILES | O[C@H]1[C@@H](Cc2cccnc2)COc2ccc(OCc3ccc4ccccc4n3)cc12 |
|---|
| Structure | 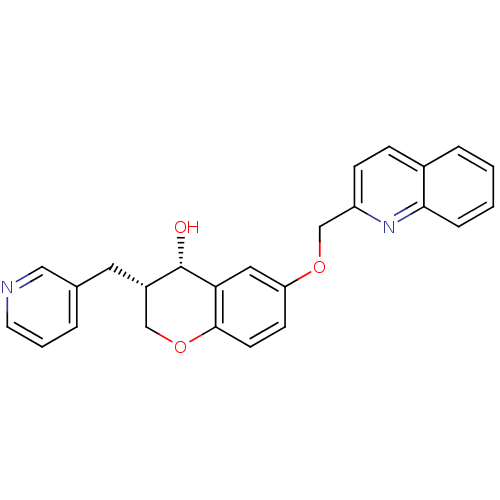
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Andrews, EG; Antognoli, GW; Breslow, R; Carta, MP; Carty, TJ; Chambers, RJ; Cheng, JB; Cohan, VL; Collins, JL; Damon, DB; Delehunt, J; Eggler, JF; Eskra, JD; Freiert, KW; Hada, WA; Marfat, A; Masamune, H; Melvin, LS; Mularski, CJ; Naclerio, BA Synthesis and pharmacological profile of two novel heterocyclic chromanols, CP-80,798 and CP-85,958, as potent LTD4 receptor antagonists Bioorg Med Chem Lett5:1365-1370 (1995) Article
Andrews, EG; Antognoli, GW; Breslow, R; Carta, MP; Carty, TJ; Chambers, RJ; Cheng, JB; Cohan, VL; Collins, JL; Damon, DB; Delehunt, J; Eggler, JF; Eskra, JD; Freiert, KW; Hada, WA; Marfat, A; Masamune, H; Melvin, LS; Mularski, CJ; Naclerio, BA Synthesis and pharmacological profile of two novel heterocyclic chromanols, CP-80,798 and CP-85,958, as potent LTD4 receptor antagonists Bioorg Med Chem Lett5:1365-1370 (1995) Article