| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50302371 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_597146 (CHEMBL1048901) |
|---|
| IC50 | 48000±n/a nM |
|---|
| Citation |  Giblin, GM; Billinton, A; Briggs, M; Brown, AJ; Chessell, IP; Clayton, NM; Eatherton, AJ; Goldsmith, P; Haslam, C; Johnson, MR; Mitchell, WL; Naylor, A; Perboni, A; Slingsby, BP; Wilson, AW Discovery of 1-[4-(3-chlorophenylamino)-1-methyl-1H-pyrrolo[3,2-c]pyridin-7-yl]-1-morpholin-4-ylmethanone (GSK554418A), a brain penetrant 5-azaindole CB2 agonist for the treatment of chronic pain. J Med Chem52:5785-8 (2009) [PubMed] Article Giblin, GM; Billinton, A; Briggs, M; Brown, AJ; Chessell, IP; Clayton, NM; Eatherton, AJ; Goldsmith, P; Haslam, C; Johnson, MR; Mitchell, WL; Naylor, A; Perboni, A; Slingsby, BP; Wilson, AW Discovery of 1-[4-(3-chlorophenylamino)-1-methyl-1H-pyrrolo[3,2-c]pyridin-7-yl]-1-morpholin-4-ylmethanone (GSK554418A), a brain penetrant 5-azaindole CB2 agonist for the treatment of chronic pain. J Med Chem52:5785-8 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50302371 |
|---|
| n/a |
|---|
| Name | BDBM50302371 |
|---|
| Synonyms: | 1-[7-(3-Chloro-phenylamino)-3-methyl-1H-pyrrolo[2,3-c]pyridin-4-yl]-1-morpholin-4-yl-methanone | CHEMBL567911 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H19ClN4O2 |
|---|
| Mol. Mass. | 370.833 |
|---|
| SMILES | Cc1c[nH]c2c(Nc3cccc(Cl)c3)ncc(C(=O)N3CCOCC3)c12 |
|---|
| Structure | 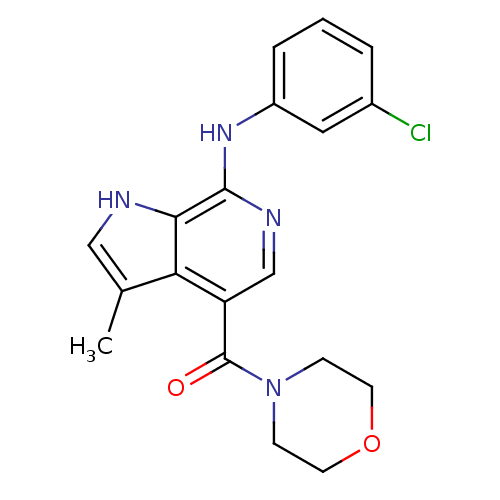
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Giblin, GM; Billinton, A; Briggs, M; Brown, AJ; Chessell, IP; Clayton, NM; Eatherton, AJ; Goldsmith, P; Haslam, C; Johnson, MR; Mitchell, WL; Naylor, A; Perboni, A; Slingsby, BP; Wilson, AW Discovery of 1-[4-(3-chlorophenylamino)-1-methyl-1H-pyrrolo[3,2-c]pyridin-7-yl]-1-morpholin-4-ylmethanone (GSK554418A), a brain penetrant 5-azaindole CB2 agonist for the treatment of chronic pain. J Med Chem52:5785-8 (2009) [PubMed] Article
Giblin, GM; Billinton, A; Briggs, M; Brown, AJ; Chessell, IP; Clayton, NM; Eatherton, AJ; Goldsmith, P; Haslam, C; Johnson, MR; Mitchell, WL; Naylor, A; Perboni, A; Slingsby, BP; Wilson, AW Discovery of 1-[4-(3-chlorophenylamino)-1-methyl-1H-pyrrolo[3,2-c]pyridin-7-yl]-1-morpholin-4-ylmethanone (GSK554418A), a brain penetrant 5-azaindole CB2 agonist for the treatment of chronic pain. J Med Chem52:5785-8 (2009) [PubMed] Article