| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene A-4 hydrolase |
|---|
| Ligand | BDBM50303657 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_596628 (CHEMBL1046155) |
|---|
| IC50 | 23±n/a nM |
|---|
| Citation |  Sandanayaka, V; Mamat, B; Mishra, RK; Winger, J; Krohn, M; Zhou, LM; Keyvan, M; Enache, L; Sullins, D; Onua, E; Zhang, J; Halldorsdottir, G; Sigthorsdottir, H; Thorlaksdottir, A; Sigthorsson, G; Thorsteinnsdottir, M; Davies, DR; Stewart, LJ; Zembower, DE; Andresson, T; Kiselyov, AS; Singh, J; Gurney, ME Discovery of 4-[(2S)-2-{[4-(4-chlorophenoxy)phenoxy]methyl}-1-pyrrolidinyl]butanoic acid (DG-051) as a novel leukotriene A4 hydrolase inhibitor of leukotriene B4 biosynthesis. J Med Chem53:573-85 (2010) [PubMed] Article Sandanayaka, V; Mamat, B; Mishra, RK; Winger, J; Krohn, M; Zhou, LM; Keyvan, M; Enache, L; Sullins, D; Onua, E; Zhang, J; Halldorsdottir, G; Sigthorsdottir, H; Thorlaksdottir, A; Sigthorsson, G; Thorsteinnsdottir, M; Davies, DR; Stewart, LJ; Zembower, DE; Andresson, T; Kiselyov, AS; Singh, J; Gurney, ME Discovery of 4-[(2S)-2-{[4-(4-chlorophenoxy)phenoxy]methyl}-1-pyrrolidinyl]butanoic acid (DG-051) as a novel leukotriene A4 hydrolase inhibitor of leukotriene B4 biosynthesis. J Med Chem53:573-85 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Leukotriene A-4 hydrolase |
|---|
| Name: | Leukotriene A-4 hydrolase |
|---|
| Synonyms: | LKHA4_HUMAN | LTA-4 hydrolase | LTA4 | LTA4H | Leukotriene A(4) hydrolase | Leukotriene A-4 hydrolase (LTA4H) | Leukotriene A4 hydrolase |
|---|
| Type: | Hydrolase; metalloprotease |
|---|
| Mol. Mass.: | 69280.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant LTA4H. |
|---|
| Residue: | 611 |
|---|
| Sequence: | MPEIVDTCSLASPASVCRTKHLHLRCSVDFTRRTLTGTAALTVQSQEDNLRSLVLDTKDL
TIEKVVINGQEVKYALGERQSYKGSPMEISLPIALSKNQEIVIEISFETSPKSSALQWLT
PEQTSGKEHPYLFSQCQAIHCRAILPCQDTPSVKLTYTAEVSVPKELVALMSAIRDGETP
DPEDPSRKIYKFIQKVPIPCYLIALVVGALESRQIGPRTLVWSEKEQVEKSAYEFSETES
MLKIAEDLGGPYVWGQYDLLVLPPSFPYGGMENPCLTFVTPTLLAGDKSLSNVIAHEISH
SWTGNLVTNKTWDHFWLNEGHTVYLERHICGRLFGEKFRHFNALGGWGELQNSVKTFGET
HPFTKLVVDLTDIDPDVAYSSVPYEKGFALLFYLEQLLGGPEIFLGFLKAYVEKFSYKSI
TTDDWKDFLYSYFKDKVDVLNQVDWNAWLYSPGLPPIKPNYDMTLTNACIALSQRWITAK
EDDLNSFNATDLKDLSSHQLNEFLAQTLQRAPLPLGHIKRMQEVYNFNAINNSEIRFRWL
RLCIQSKWEDAIPLALKMATEQGRMKFTRPLFKDLAAFDKSHDQAVRTYQEHKASMHPVT
AMLVGKDLKVD
|
|
|
|---|
| BDBM50303657 |
|---|
| n/a |
|---|
| Name | BDBM50303657 |
|---|
| Synonyms: | 3-{(R)-2-[4-(4-Chlorophenoxy)phenoxymethyl]pyrrolidin-1-yl}-propionic Acid | CHEMBL565605 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H22ClNO4 |
|---|
| Mol. Mass. | 375.846 |
|---|
| SMILES | OC(=O)CCN1CCC[C@@H]1COc1ccc(Oc2ccc(Cl)cc2)cc1 |r| |
|---|
| Structure | 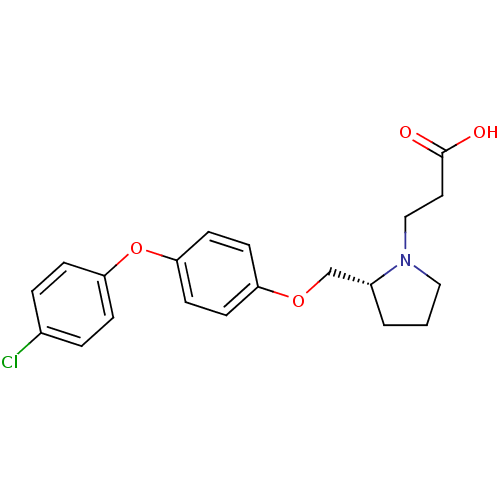
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sandanayaka, V; Mamat, B; Mishra, RK; Winger, J; Krohn, M; Zhou, LM; Keyvan, M; Enache, L; Sullins, D; Onua, E; Zhang, J; Halldorsdottir, G; Sigthorsdottir, H; Thorlaksdottir, A; Sigthorsson, G; Thorsteinnsdottir, M; Davies, DR; Stewart, LJ; Zembower, DE; Andresson, T; Kiselyov, AS; Singh, J; Gurney, ME Discovery of 4-[(2S)-2-{[4-(4-chlorophenoxy)phenoxy]methyl}-1-pyrrolidinyl]butanoic acid (DG-051) as a novel leukotriene A4 hydrolase inhibitor of leukotriene B4 biosynthesis. J Med Chem53:573-85 (2010) [PubMed] Article
Sandanayaka, V; Mamat, B; Mishra, RK; Winger, J; Krohn, M; Zhou, LM; Keyvan, M; Enache, L; Sullins, D; Onua, E; Zhang, J; Halldorsdottir, G; Sigthorsdottir, H; Thorlaksdottir, A; Sigthorsson, G; Thorsteinnsdottir, M; Davies, DR; Stewart, LJ; Zembower, DE; Andresson, T; Kiselyov, AS; Singh, J; Gurney, ME Discovery of 4-[(2S)-2-{[4-(4-chlorophenoxy)phenoxy]methyl}-1-pyrrolidinyl]butanoic acid (DG-051) as a novel leukotriene A4 hydrolase inhibitor of leukotriene B4 biosynthesis. J Med Chem53:573-85 (2010) [PubMed] Article