| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50306148 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_606078 (CHEMBL1071977) |
|---|
| Ki | 1.5±n/a nM |
|---|
| Citation |  Kleanthous, S; Borthwick, AD; Brown, D; Burns-Kurtis, CL; Campbell, M; Chaudry, L; Chan, C; Clarte, MO; Convery, MA; Harling, JD; Hortense, E; Irving, WR; Irvine, S; Pateman, AJ; Patikis, AN; Pinto, IL; Pollard, DR; Roethka, TJ; Senger, S; Shah, GP; Stelman, GJ; Toomey, JR; Watson, NS; West, RI; Whittaker, C; Zhou, P; Young, RJ Structure and property based design of factor Xa inhibitors: pyrrolidin-2-ones with monoaryl P4 motifs. Bioorg Med Chem Lett20:618-22 (2010) [PubMed] Article Kleanthous, S; Borthwick, AD; Brown, D; Burns-Kurtis, CL; Campbell, M; Chaudry, L; Chan, C; Clarte, MO; Convery, MA; Harling, JD; Hortense, E; Irving, WR; Irvine, S; Pateman, AJ; Patikis, AN; Pinto, IL; Pollard, DR; Roethka, TJ; Senger, S; Shah, GP; Stelman, GJ; Toomey, JR; Watson, NS; West, RI; Whittaker, C; Zhou, P; Young, RJ Structure and property based design of factor Xa inhibitors: pyrrolidin-2-ones with monoaryl P4 motifs. Bioorg Med Chem Lett20:618-22 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50306148 |
|---|
| n/a |
|---|
| Name | BDBM50306148 |
|---|
| Synonyms: | (S)-4-(3-(2-(5-chlorothiophen-2-yl)vinylsulfonamido)-2-oxopyrrolidin-1-yl)-3-fluoro-N,N-dimethylbenzamide | CHEMBL607085 | GTC000101A |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H19ClFN3O4S2 |
|---|
| Mol. Mass. | 471.953 |
|---|
| SMILES | CN(C)C(=O)c1ccc(N2CC[C@H](NS(=O)(=O)\C=C\c3ccc(Cl)s3)C2=O)c(F)c1 |r| |
|---|
| Structure | 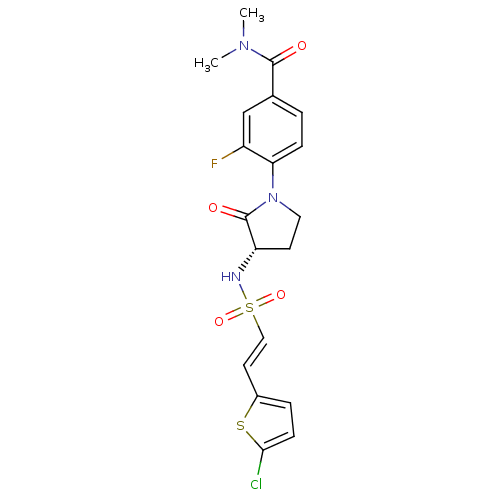
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kleanthous, S; Borthwick, AD; Brown, D; Burns-Kurtis, CL; Campbell, M; Chaudry, L; Chan, C; Clarte, MO; Convery, MA; Harling, JD; Hortense, E; Irving, WR; Irvine, S; Pateman, AJ; Patikis, AN; Pinto, IL; Pollard, DR; Roethka, TJ; Senger, S; Shah, GP; Stelman, GJ; Toomey, JR; Watson, NS; West, RI; Whittaker, C; Zhou, P; Young, RJ Structure and property based design of factor Xa inhibitors: pyrrolidin-2-ones with monoaryl P4 motifs. Bioorg Med Chem Lett20:618-22 (2010) [PubMed] Article
Kleanthous, S; Borthwick, AD; Brown, D; Burns-Kurtis, CL; Campbell, M; Chaudry, L; Chan, C; Clarte, MO; Convery, MA; Harling, JD; Hortense, E; Irving, WR; Irvine, S; Pateman, AJ; Patikis, AN; Pinto, IL; Pollard, DR; Roethka, TJ; Senger, S; Shah, GP; Stelman, GJ; Toomey, JR; Watson, NS; West, RI; Whittaker, C; Zhou, P; Young, RJ Structure and property based design of factor Xa inhibitors: pyrrolidin-2-ones with monoaryl P4 motifs. Bioorg Med Chem Lett20:618-22 (2010) [PubMed] Article