| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase receptor UFO |
|---|
| Ligand | BDBM50306677 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_611547 (CHEMBL1066391) |
|---|
| IC50 | 570±n/a nM |
|---|
| Citation |  Nishii, H; Chiba, T; Morikami, K; Fukami, TA; Sakamoto, H; Ko, K; Koyano, H Discovery of 6-benzyloxyquinolines as c-Met selective kinase inhibitors. Bioorg Med Chem Lett20:1405-9 (2010) [PubMed] Article Nishii, H; Chiba, T; Morikami, K; Fukami, TA; Sakamoto, H; Ko, K; Koyano, H Discovery of 6-benzyloxyquinolines as c-Met selective kinase inhibitors. Bioorg Med Chem Lett20:1405-9 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase receptor UFO |
|---|
| Name: | Tyrosine-protein kinase receptor UFO |
|---|
| Synonyms: | AXL | AXL oncogene | TEL/AXL | Tyrosine-protein kinase receptor UFO (AXL) | UFO | UFO_HUMAN |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 98316.97 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P30530 |
|---|
| Residue: | 894 |
|---|
| Sequence: | MAWRCPRMGRVPLAWCLALCGWACMAPRGTQAEESPFVGNPGNITGARGLTGTLRCQLQV
QGEPPEVHWLRDGQILELADSTQTQVPLGEDEQDDWIVVSQLRITSLQLSDTGQYQCLVF
LGHQTFVSQPGYVGLEGLPYFLEEPEDRTVAANTPFNLSCQAQGPPEPVDLLWLQDAVPL
ATAPGHGPQRSLHVPGLNKTSSFSCEAHNAKGVTTSRTATITVLPQQPRNLHLVSRQPTE
LEVAWTPGLSGIYPLTHCTLQAVLSDDGMGIQAGEPDPPEEPLTSQASVPPHQLRLGSLH
PHTPYHIRVACTSSQGPSSWTHWLPVETPEGVPLGPPENISATRNGSQAFVHWQEPRAPL
QGTLLGYRLAYQGQDTPEVLMDIGLRQEVTLELQGDGSVSNLTVCVAAYTAAGDGPWSLP
VPLEAWRPGQAQPVHQLVKEPSTPAFSWPWWYVLLGAVVAAACVLILALFLVHRRKKETR
YGEVFEPTVERGELVVRYRVRKSYSRRTTEATLNSLGISEELKEKLRDVMVDRHKVALGK
TLGEGEFGAVMEGQLNQDDSILKVAVKTMKIAICTRSELEDFLSEAVCMKEFDHPNVMRL
IGVCFQGSERESFPAPVVILPFMKHGDLHSFLLYSRLGDQPVYLPTQMLVKFMADIASGM
EYLSTKRFIHRDLAARNCMLNENMSVCVADFGLSKKIYNGDYYRQGRIAKMPVKWIAIES
LADRVYTSKSDVWSFGVTMWEIATRGQTPYPGVENSEIYDYLRQGNRLKQPADCLDGLYA
LMSRCWELNPQDRPSFTELREDLENTLKALPPAQEPDEILYVNMDEGGGYPEPPGAAGGA
DPPTQPDPKDSCSCLTAAEVHPAGRYVLCPSTTPSPAQPADRGSPAAPGQEDGA
|
|
|
|---|
| BDBM50306677 |
|---|
| n/a |
|---|
| Name | BDBM50306677 |
|---|
| Synonyms: | (R)-3-(6-(1-(2,6-dichloro-3-fluorophenyl)ethoxy)quinolin-3-yl)acrylamide | CHEMBL602471 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H15Cl2FN2O2 |
|---|
| Mol. Mass. | 405.25 |
|---|
| SMILES | C[C@@H](Oc1ccc2ncc(\C=C\C(N)=O)cc2c1)c1c(Cl)ccc(F)c1Cl |r| |
|---|
| Structure | 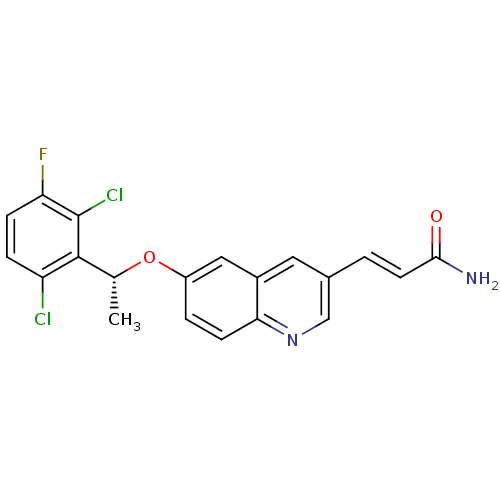
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nishii, H; Chiba, T; Morikami, K; Fukami, TA; Sakamoto, H; Ko, K; Koyano, H Discovery of 6-benzyloxyquinolines as c-Met selective kinase inhibitors. Bioorg Med Chem Lett20:1405-9 (2010) [PubMed] Article
Nishii, H; Chiba, T; Morikami, K; Fukami, TA; Sakamoto, H; Ko, K; Koyano, H Discovery of 6-benzyloxyquinolines as c-Met selective kinase inhibitors. Bioorg Med Chem Lett20:1405-9 (2010) [PubMed] Article