| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Matrix metalloproteinase-9 |
|---|
| Ligand | BDBM50314224 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_624094 (CHEMBL1103425) |
|---|
| IC50 | 13±n/a nM |
|---|
| Citation |  Nuti, E; Casalini, F; Avramova, SI; Santamaria, S; Fabbi, M; Ferrini, S; Marinelli, L; La Pietra, V; Limongelli, V; Novellino, E; Cercignani, G; Orlandini, E; Nencetti, S; Rossello, A Potent arylsulfonamide inhibitors of tumor necrosis factor-alpha converting enzyme able to reduce activated leukocyte cell adhesion molecule shedding in cancer cell models. J Med Chem53:2622-35 (2010) [PubMed] Article Nuti, E; Casalini, F; Avramova, SI; Santamaria, S; Fabbi, M; Ferrini, S; Marinelli, L; La Pietra, V; Limongelli, V; Novellino, E; Cercignani, G; Orlandini, E; Nencetti, S; Rossello, A Potent arylsulfonamide inhibitors of tumor necrosis factor-alpha converting enzyme able to reduce activated leukocyte cell adhesion molecule shedding in cancer cell models. J Med Chem53:2622-35 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Matrix metalloproteinase-9 |
|---|
| Name: | Matrix metalloproteinase-9 |
|---|
| Synonyms: | 67 kDa matrix metalloproteinase-9 | 82 kDa matrix metalloproteinase-9 | 92 kDa gelatinase | 92 kDa type IV collagenase | CLG4B | GELB | Gelatinase B | MMP-9 | MMP9 | MMP9_HUMAN | Matrix metalloproteinase 9 (MMP-9) | Matrix metalloproteinase-9 (MMP-9) | Matrix metalloproteinase-9 (MMP9) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 78452.28 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14780 |
|---|
| Residue: | 707 |
|---|
| Sequence: | MSLWQPLVLVLLVLGCCFAAPRQRQSTLVLFPGDLRTNLTDRQLAEEYLYRYGYTRVAEM
RGESKSLGPALLLLQKQLSLPETGELDSATLKAMRTPRCGVPDLGRFQTFEGDLKWHHHN
ITYWIQNYSEDLPRAVIDDAFARAFALWSAVTPLTFTRVYSRDADIVIQFGVAEHGDGYP
FDGKDGLLAHAFPPGPGIQGDAHFDDDELWSLGKGVVVPTRFGNADGAACHFPFIFEGRS
YSACTTDGRSDGLPWCSTTANYDTDDRFGFCPSERLYTQDGNADGKPCQFPFIFQGQSYS
ACTTDGRSDGYRWCATTANYDRDKLFGFCPTRADSTVMGGNSAGELCVFPFTFLGKEYST
CTSEGRGDGRLWCATTSNFDSDKKWGFCPDQGYSLFLVAAHEFGHALGLDHSSVPEALMY
PMYRFTEGPPLHKDDVNGIRHLYGPRPEPEPRPPTTTTPQPTAPPTVCPTGPPTVHPSER
PTAGPTGPPSAGPTGPPTAGPSTATTVPLSPVDDACNVNIFDAIAEIGNQLYLFKDGKYW
RFSEGRGSRPQGPFLIADKWPALPRKLDSVFEERLSKKLFFFSGRQVWVYTGASVLGPRR
LDKLGLGADVAQVTGALRSGRGKMLLFSGRRLWRFDVKAQMVDPRSASEVDRMFPGVPLD
THDVFQYREKAYFCQDRFYWRVSSRSELNQVDQVGYVTYDILQCPED
|
|
|
|---|
| BDBM50314224 |
|---|
| n/a |
|---|
| Name | BDBM50314224 |
|---|
| Synonyms: | (R)-2-(4-(but-2-ynyloxy)-N-isobutylphenylsulfonamido)-4-(1,3-dioxoisoindolin-2-yl)-N-hydroxybutanamide | CHEMBL1088878 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H29N3O7S |
|---|
| Mol. Mass. | 527.589 |
|---|
| SMILES | CC#CCOc1ccc(cc1)S(=O)(=O)N(CC(C)C)[C@H](CCN1C(=O)c2ccccc2C1=O)C(=O)NO |r| |
|---|
| Structure | 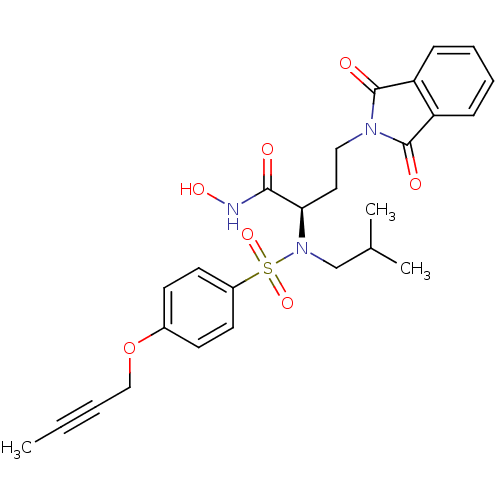
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nuti, E; Casalini, F; Avramova, SI; Santamaria, S; Fabbi, M; Ferrini, S; Marinelli, L; La Pietra, V; Limongelli, V; Novellino, E; Cercignani, G; Orlandini, E; Nencetti, S; Rossello, A Potent arylsulfonamide inhibitors of tumor necrosis factor-alpha converting enzyme able to reduce activated leukocyte cell adhesion molecule shedding in cancer cell models. J Med Chem53:2622-35 (2010) [PubMed] Article
Nuti, E; Casalini, F; Avramova, SI; Santamaria, S; Fabbi, M; Ferrini, S; Marinelli, L; La Pietra, V; Limongelli, V; Novellino, E; Cercignani, G; Orlandini, E; Nencetti, S; Rossello, A Potent arylsulfonamide inhibitors of tumor necrosis factor-alpha converting enzyme able to reduce activated leukocyte cell adhesion molecule shedding in cancer cell models. J Med Chem53:2622-35 (2010) [PubMed] Article