| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Smoothened homolog |
|---|
| Ligand | BDBM50320355 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_633891 (CHEMBL1120457) |
|---|
| IC50 | 4.4±n/a nM |
|---|
| Citation |  Lucas, BS; Aaron, W; An, S; Austin, RJ; Brown, M; Chan, H; Chong, A; Hungate, R; Huang, T; Jiang, B; Johnson, MG; Kaizerman, JA; Lee, G; McMinn, DL; Orf, J; Powers, JP; Rong, M; Toteva, MM; Uyeda, C; Wickramasinghe, D; Xu, G; Ye, Q; Zhong, W Design of 1-piperazinyl-4-arylphthalazines as potent Smoothened antagonists. Bioorg Med Chem Lett20:3618-22 (2010) [PubMed] Article Lucas, BS; Aaron, W; An, S; Austin, RJ; Brown, M; Chan, H; Chong, A; Hungate, R; Huang, T; Jiang, B; Johnson, MG; Kaizerman, JA; Lee, G; McMinn, DL; Orf, J; Powers, JP; Rong, M; Toteva, MM; Uyeda, C; Wickramasinghe, D; Xu, G; Ye, Q; Zhong, W Design of 1-piperazinyl-4-arylphthalazines as potent Smoothened antagonists. Bioorg Med Chem Lett20:3618-22 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Smoothened homolog |
|---|
| Name: | Smoothened homolog |
|---|
| Synonyms: | G-protein- coupled-like receptor Smoothened (Smo) | SMO | SMOH | SMO_HUMAN | Smoothened homolog |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 86415.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q99835 |
|---|
| Residue: | 787 |
|---|
| Sequence: | MAAARPARGPELPLLGLLLLLLLGDPGRGAASSGNATGPGPRSAGGSARRSAAVTGPPPP
LSHCGRAAPCEPLRYNVCLGSVLPYGATSTLLAGDSDSQEEAHGKLVLWSGLRNAPRCWA
VIQPLLCAVYMPKCENDRVELPSRTLCQATRGPCAIVERERGWPDFLRCTPDRFPEGCTN
EVQNIKFNSSGQCEVPLVRTDNPKSWYEDVEGCGIQCQNPLFTEAEHQDMHSYIAAFGAV
TGLCTLFTLATFVADWRNSNRYPAVILFYVNACFFVGSIGWLAQFMDGARREIVCRADGT
MRLGEPTSNETLSCVIIFVIVYYALMAGVVWFVVLTYAWHTSFKALGTTYQPLSGKTSYF
HLLTWSLPFVLTVAILAVAQVDGDSVSGICFVGYKNYRYRAGFVLAPIGLVLIVGGYFLI
RGVMTLFSIKSNHPGLLSEKAASKINETMLRLGIFGFLAFGFVLITFSCHFYDFFNQAEW
ERSFRDYVLCQANVTIGLPTKQPIPDCEIKNRPSLLVEKINLFAMFGTGIAMSTWVWTKA
TLLIWRRTWCRLTGQSDDEPKRIKKSKMIAKAFSKRHELLQNPGQELSFSMHTVSHDGPV
AGLAFDLNEPSADVSSAWAQHVTKMVARRGAILPQDISVTPVATPVPPEEQANLWLVEAE
ISPELQKRLGRKKKRRKRKKEVCPLAPPPELHPPAPAPSTIPRLPQLPRQKCLVAAGAWG
AGDSCRQGAWTLVSNPFCPEPSPPQDPFLPSAPAPVAWAHGRRQGLGPIHSRTNLMDTEL
MDADSDF
|
|
|
|---|
| BDBM50320355 |
|---|
| n/a |
|---|
| Name | BDBM50320355 |
|---|
| Synonyms: | CHEMBL1085503 | phenyl(4-(4-p-tolylphthalazin-1-yl)piperazin-1-yl)methanone |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H24N4O |
|---|
| Mol. Mass. | 408.495 |
|---|
| SMILES | Cc1ccc(cc1)-c1nnc(N2CCN(CC2)C(=O)c2ccccc2)c2ccccc12 |
|---|
| Structure | 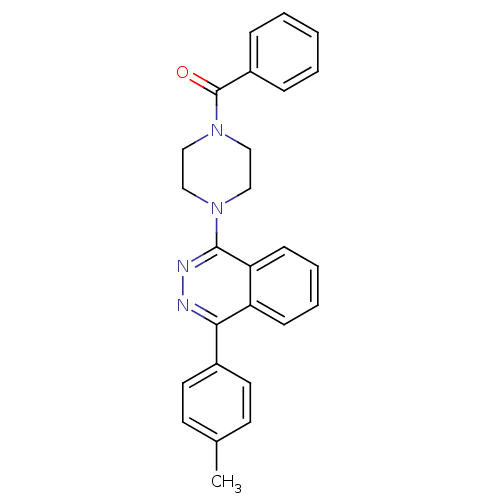
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lucas, BS; Aaron, W; An, S; Austin, RJ; Brown, M; Chan, H; Chong, A; Hungate, R; Huang, T; Jiang, B; Johnson, MG; Kaizerman, JA; Lee, G; McMinn, DL; Orf, J; Powers, JP; Rong, M; Toteva, MM; Uyeda, C; Wickramasinghe, D; Xu, G; Ye, Q; Zhong, W Design of 1-piperazinyl-4-arylphthalazines as potent Smoothened antagonists. Bioorg Med Chem Lett20:3618-22 (2010) [PubMed] Article
Lucas, BS; Aaron, W; An, S; Austin, RJ; Brown, M; Chan, H; Chong, A; Hungate, R; Huang, T; Jiang, B; Johnson, MG; Kaizerman, JA; Lee, G; McMinn, DL; Orf, J; Powers, JP; Rong, M; Toteva, MM; Uyeda, C; Wickramasinghe, D; Xu, G; Ye, Q; Zhong, W Design of 1-piperazinyl-4-arylphthalazines as potent Smoothened antagonists. Bioorg Med Chem Lett20:3618-22 (2010) [PubMed] Article