

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 2C9 | ||
| Ligand | BDBM50323161 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_643849 (CHEMBL1211748) | ||
| IC50 | >40000±n/a nM | ||
| Citation |  Brigance, RP; Meng, W; Fura, A; Harrity, T; Wang, A; Zahler, R; Kirby, MS; Hamann, LG Synthesis and SAR of azolopyrimidines as potent and selective dipeptidyl peptidase-4 (DPP4) inhibitors for type 2 diabetes. Bioorg Med Chem Lett20:4395-8 (2010) [PubMed] Article Brigance, RP; Meng, W; Fura, A; Harrity, T; Wang, A; Zahler, R; Kirby, MS; Hamann, LG Synthesis and SAR of azolopyrimidines as potent and selective dipeptidyl peptidase-4 (DPP4) inhibitors for type 2 diabetes. Bioorg Med Chem Lett20:4395-8 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 2C9 | |||
| Name: | Cytochrome P450 2C9 | ||
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 55636.33 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P11712 | ||
| Residue: | 490 | ||
| Sequence: |
| ||
| BDBM50323161 | |||
| n/a | |||
| Name | BDBM50323161 | ||
| Synonyms: | (5-(2,4-dichlorophenyl)-7-methyl-2-phenylimidazo[1,2-a]pyrimidin-6-yl)methanamine | CHEMBL1209853 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C20H16Cl2N4 | ||
| Mol. Mass. | 383.274 | ||
| SMILES | Cc1nc2nc(cn2c(c1CN)-c1ccc(Cl)cc1Cl)-c1ccccc1 |(19.44,4.47,;18.11,3.7,;16.77,4.47,;15.43,3.69,;13.96,4.16,;13.06,2.9,;13.98,1.66,;15.45,2.15,;16.78,1.39,;18.11,2.16,;19.45,1.39,;20.78,2.17,;16.79,-.15,;15.46,-.92,;15.46,-2.46,;16.79,-3.23,;16.79,-4.77,;18.13,-2.46,;18.12,-.91,;19.45,-.14,;11.53,2.89,;10.75,4.22,;9.21,4.21,;8.45,2.88,;9.23,1.54,;10.77,1.56,)| | ||
| Structure | 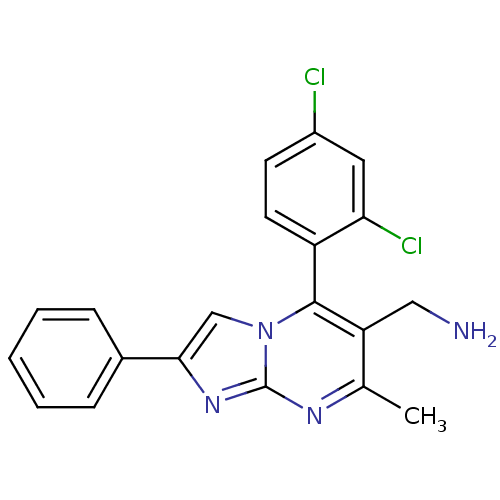
| ||