| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50325460 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_652208 (CHEMBL1228481) |
|---|
| IC50 | 400±n/a nM |
|---|
| Citation |  Ting, PC; Lee, JF; Albanese, MM; Wu, J; Aslanian, R; Favreau, L; Nardo, C; Korfmacher, WA; West, RE; Williams, SM; Anthes, JC; Rivelli, MA; Corboz, MR; Hey, JA The synthesis and structure-activity relationship of 4-benzimidazolyl-piperidinylcarbonyl-piperidine analogs as histamine H3 antagonists. Bioorg Med Chem Lett20:5004-8 (2010) [PubMed] Article Ting, PC; Lee, JF; Albanese, MM; Wu, J; Aslanian, R; Favreau, L; Nardo, C; Korfmacher, WA; West, RE; Williams, SM; Anthes, JC; Rivelli, MA; Corboz, MR; Hey, JA The synthesis and structure-activity relationship of 4-benzimidazolyl-piperidinylcarbonyl-piperidine analogs as histamine H3 antagonists. Bioorg Med Chem Lett20:5004-8 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50325460 |
|---|
| n/a |
|---|
| Name | BDBM50325460 |
|---|
| Synonyms: | 1-(4-methoxybenzyl)-3-(1-(1-(pyridin-4-ylmethyl)piperidine-4-carbonyl)piperidin-4-yl)-1H-benzo[d]imidazol-2(3H)-one | CHEMBL1223003 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H37N5O3 |
|---|
| Mol. Mass. | 539.6679 |
|---|
| SMILES | COc1ccc(Cn2c3ccccc3n(C3CCN(CC3)C(=O)C3CCN(Cc4ccncc4)CC3)c2=O)cc1 |
|---|
| Structure | 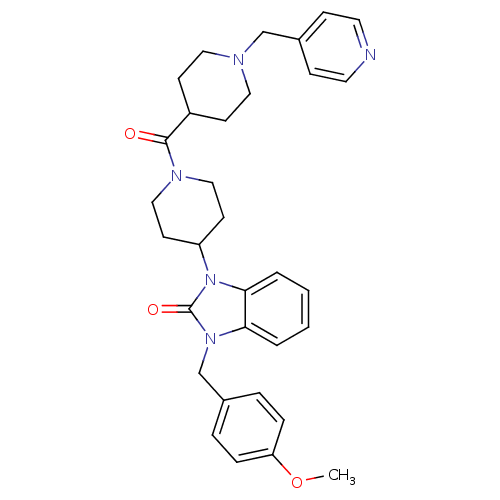
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ting, PC; Lee, JF; Albanese, MM; Wu, J; Aslanian, R; Favreau, L; Nardo, C; Korfmacher, WA; West, RE; Williams, SM; Anthes, JC; Rivelli, MA; Corboz, MR; Hey, JA The synthesis and structure-activity relationship of 4-benzimidazolyl-piperidinylcarbonyl-piperidine analogs as histamine H3 antagonists. Bioorg Med Chem Lett20:5004-8 (2010) [PubMed] Article
Ting, PC; Lee, JF; Albanese, MM; Wu, J; Aslanian, R; Favreau, L; Nardo, C; Korfmacher, WA; West, RE; Williams, SM; Anthes, JC; Rivelli, MA; Corboz, MR; Hey, JA The synthesis and structure-activity relationship of 4-benzimidazolyl-piperidinylcarbonyl-piperidine analogs as histamine H3 antagonists. Bioorg Med Chem Lett20:5004-8 (2010) [PubMed] Article