| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium/glucose cotransporter 2 |
|---|
| Ligand | BDBM50326284 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_659926 (CHEMBL1247184) |
|---|
| IC50 | 2.2±n/a nM |
|---|
| Citation |  Nomura, S; Sakamaki, S; Hongu, M; Kawanishi, E; Koga, Y; Sakamoto, T; Yamamoto, Y; Ueta, K; Kimata, H; Nakayama, K; Tsuda-Tsukimoto, M Discovery of canagliflozin, a novel C-glucoside with thiophene ring, as sodium-dependent glucose cotransporter 2 inhibitor for the treatment of type 2 diabetes mellitus. J Med Chem53:6355-60 (2010) [PubMed] Article Nomura, S; Sakamaki, S; Hongu, M; Kawanishi, E; Koga, Y; Sakamoto, T; Yamamoto, Y; Ueta, K; Kimata, H; Nakayama, K; Tsuda-Tsukimoto, M Discovery of canagliflozin, a novel C-glucoside with thiophene ring, as sodium-dependent glucose cotransporter 2 inhibitor for the treatment of type 2 diabetes mellitus. J Med Chem53:6355-60 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium/glucose cotransporter 2 |
|---|
| Name: | Sodium/glucose cotransporter 2 |
|---|
| Synonyms: | Na(+)/glucose cotransporter 2 | SC5A2_HUMAN | SGLT2 | SLC5A2 | Sodium-Dependent Glucose Cotransporter 2 (SGLT2) | Sodium/glucose cotransporter 1 (SGLT1) | Solute carrier family 5 member 2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 72902.00 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P31639 |
|---|
| Residue: | 672 |
|---|
| Sequence: | MEEHTEAGSAPEMGAQKALIDNPADILVIAAYFLLVIGVGLWSMCRTNRGTVGGYFLAGR
SMVWWPVGASLFASNIGSGHFVGLAGTGAASGLAVAGFEWNALFVVLLLGWLFAPVYLTA
GVITMPQYLRKRFGGRRIRLYLSVLSLFLYIFTKISVDMFSGAVFIQQALGWNIYASVIA
LLGITMIYTVTGGLAALMYTDTVQTFVILGGACILMGYAFHEVGGYSGLFDKYLGAATSL
TVSEDPAVGNISSFCYRPRPDSYHLLRHPVTGDLPWPALLLGLTIVSGWYWCSDQVIVQR
CLAGKSLTHIKAGCILCGYLKLTPMFLMVMPGMISRILYPDEVACVVPEVCRRVCGTEVG
CSNIAYPRLVVKLMPNGLRGLMLAVMLAALMSSLASIFNSSSTLFTMDIYTRLRPRAGDR
ELLLVGRLWVVFIVVVSVAWLPVVQAAQGGQLFDYIQAVSSYLAPPVSAVFVLALFVPRV
NEQGAFWGLIGGLLMGLARLIPEFSFGSGSCVQPSACPAFLCGVHYLYFAIVLFFCSGLL
TLTVSLCTAPIPRKHLHRLVFSLRHSKEEREDLDADEQQGSSLPVQNGCPESAMEMNEPQ
APAPSLFRQCLLWFCGMSRGGVGSPPPLTQEEAAAAARRLEDISEDPSWARVVNLNALLM
MAVAVFLWGFYA
|
|
|
|---|
| BDBM50326284 |
|---|
| n/a |
|---|
| Name | BDBM50326284 |
|---|
| Synonyms: | 1-(beta-D-Glucopyranosyl)-4-methyl-3-(5-(4-fluorophenyl)-2-thienylmethyl)benzene | CHEMBL1243275 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H25FO6S |
|---|
| Mol. Mass. | 460.515 |
|---|
| SMILES | Cc1cccc(O[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)c1Cc1ccc(s1)-c1ccc(F)cc1 |r| |
|---|
| Structure | 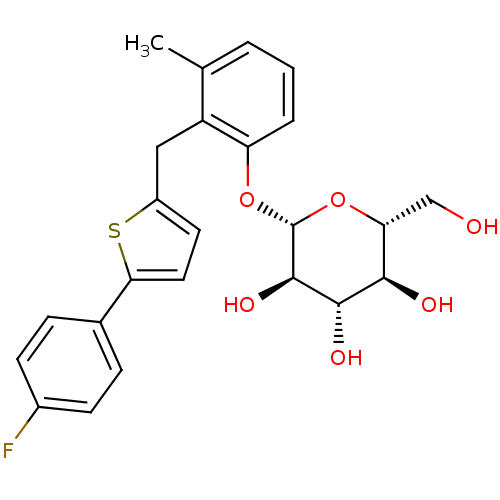
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nomura, S; Sakamaki, S; Hongu, M; Kawanishi, E; Koga, Y; Sakamoto, T; Yamamoto, Y; Ueta, K; Kimata, H; Nakayama, K; Tsuda-Tsukimoto, M Discovery of canagliflozin, a novel C-glucoside with thiophene ring, as sodium-dependent glucose cotransporter 2 inhibitor for the treatment of type 2 diabetes mellitus. J Med Chem53:6355-60 (2010) [PubMed] Article
Nomura, S; Sakamaki, S; Hongu, M; Kawanishi, E; Koga, Y; Sakamoto, T; Yamamoto, Y; Ueta, K; Kimata, H; Nakayama, K; Tsuda-Tsukimoto, M Discovery of canagliflozin, a novel C-glucoside with thiophene ring, as sodium-dependent glucose cotransporter 2 inhibitor for the treatment of type 2 diabetes mellitus. J Med Chem53:6355-60 (2010) [PubMed] Article