| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA gyrase subunit B |
|---|
| Ligand | BDBM50423645 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_677264 (CHEMBL1279102) |
|---|
| IC50 | 200±n/a nM |
|---|
| Citation |  Anderle, C; Stieger, M; Burrell, M; Reinelt, S; Maxwell, A; Page, M; Heide, L Biological activities of novel gyrase inhibitors of the aminocoumarin class. Antimicrob Agents Chemother52:1982-90 (2008) [PubMed] Article Anderle, C; Stieger, M; Burrell, M; Reinelt, S; Maxwell, A; Page, M; Heide, L Biological activities of novel gyrase inhibitors of the aminocoumarin class. Antimicrob Agents Chemother52:1982-90 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| DNA gyrase subunit B |
|---|
| Name: | DNA gyrase subunit B |
|---|
| Synonyms: | DNA gyrase subunit B | DNA gyrase subunit B (gyrB) | GYRB_ECOLI | Type IIA topoisomerase subunit GyrB | acrB | cou | gyrB | himB | hisU | nalC | parA | pcbA |
|---|
| Type: | Enzyme Subunit |
|---|
| Mol. Mass.: | 89941.28 |
|---|
| Organism: | Escherichia coli (strain K12) |
|---|
| Description: | P0AES6 |
|---|
| Residue: | 804 |
|---|
| Sequence: | MSNSYDSSSIKVLKGLDAVRKRPGMYIGDTDDGTGLHHMVFEVVDNAIDEALAGHCKEII
VTIHADNSVSVQDDGRGIPTGIHPEEGVSAAEVIMTVLHAGGKFDDNSYKVSGGLHGVGV
SVVNALSQKLELVIQREGKIHRQIYEHGVPQAPLAVTGETEKTGTMVRFWPSLETFTNVT
EFEYEILAKRLRELSFLNSGVSIRLRDKRDGKEDHFHYEGGIKAFVEYLNKNKTPIHPNI
FYFSTEKDGIGVEVALQWNDGFQENIYCFTNNIPQRDGGTHLAGFRAAMTRTLNAYMDKE
GYSKKAKVSATGDDAREGLIAVVSVKVPDPKFSSQTKDKLVSSEVKSAVEQQMNELLAEY
LLENPTDAKIVVGKIIDAARAREAARRAREMTRRKGALDLAGLPGKLADCQERDPALSEL
YLVEGDSAGGSAKQGRNRKNQAILPLKGKILNVEKARFDKMLSSQEVATLITALGCGIGR
DEYNPDKLRYHSIIIMTDADVDGSHIRTLLLTFFYRQMPEIVERGHVYIAQPPLYKVKKG
KQEQYIKDDEAMDQYQISIALDGATLHTNASAPALAGEALEKLVSEYNATQKMINRMERR
YPKAMLKELIYQPTLTEADLSDEQTVTRWVNALVSELNDKEQHGSQWKFDVHTNAEQNLF
EPIVRVRTHGVDTDYPLDHEFITGGEYRRICTLGEKLRGLLEEDAFIERGERRQPVASFE
QALDWLVKESRRGLSIQRYKGLGEMNPEQLWETTMDPESRRMLRVTVKDAIAADQLFTTL
MGDAVEPRRAFIEENALKAANIDI
|
|
|
|---|
| BDBM50423645 |
|---|
| n/a |
|---|
| Name | BDBM50423645 |
|---|
| Synonyms: | Albamycin | Cathomycin | NOVOBIOCIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H36N2O11 |
|---|
| Mol. Mass. | 612.6243 |
|---|
| SMILES | [#6]-[#8]-[#6@@H]1-[#6@@H](-[#8]-[#6](-[#7])=O)-[#6@H](-[#8])-[#6@@H](-[#8]-c2ccc3c(-[#8])c(-[#7]-[#6](=O)-c4ccc(-[#8])c(-[#6]\[#6]=[#6](\[#6])-[#6])c4)c(=O)oc3c2-[#6])-[#8]C1([#6])[#6] |
|---|
| Structure | 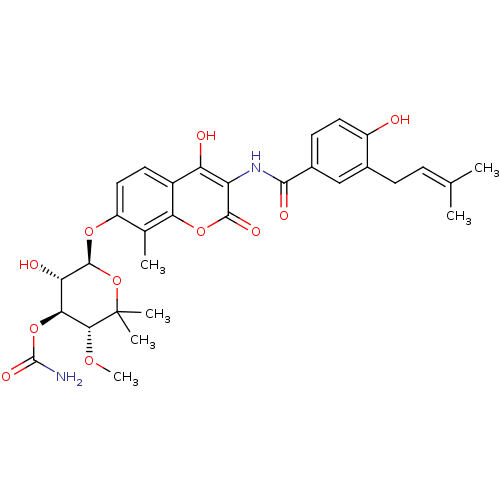
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Anderle, C; Stieger, M; Burrell, M; Reinelt, S; Maxwell, A; Page, M; Heide, L Biological activities of novel gyrase inhibitors of the aminocoumarin class. Antimicrob Agents Chemother52:1982-90 (2008) [PubMed] Article
Anderle, C; Stieger, M; Burrell, M; Reinelt, S; Maxwell, A; Page, M; Heide, L Biological activities of novel gyrase inhibitors of the aminocoumarin class. Antimicrob Agents Chemother52:1982-90 (2008) [PubMed] Article