| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prolyl endopeptidase FAP |
|---|
| Ligand | BDBM50338435 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_728293 (CHEMBL1687450) |
|---|
| IC50 | >35000±n/a nM |
|---|
| Citation |  Chen, P; Caldwell, CG; Ashton, W; Wu, JK; He, H; Lyons, KA; Thornberry, NA; Weber, AE Synthesis and evaluation of [(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]cyclohexanes and 4-[(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]piperidines as DPP-4 inhibitors. Bioorg Med Chem Lett21:1880-6 (2011) [PubMed] Article Chen, P; Caldwell, CG; Ashton, W; Wu, JK; He, H; Lyons, KA; Thornberry, NA; Weber, AE Synthesis and evaluation of [(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]cyclohexanes and 4-[(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]piperidines as DPP-4 inhibitors. Bioorg Med Chem Lett21:1880-6 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prolyl endopeptidase FAP |
|---|
| Name: | Prolyl endopeptidase FAP |
|---|
| Synonyms: | 170 kDa melanoma membrane-bound gelatinase | FAP | Fibroblast Activation Protein (FAP) | Fibroblast activation protein alpha | Integral membrane serine protease | SEPR_HUMAN | Seprase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 87712.48 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q12884 |
|---|
| Residue: | 760 |
|---|
| Sequence: | MKTWVKIVFGVATSAVLALLVMCIVLRPSRVHNSEENTMRALTLKDILNGTFSYKTFFPN
WISGQEYLHQSADNNIVLYNIETGQSYTILSNRTMKSVNASNYGLSPDRQFVYLESDYSK
LWRYSYTATYYIYDLSNGEFVRGNELPRPIQYLCWSPVGSKLAYVYQNNIYLKQRPGDPP
FQITFNGRENKIFNGIPDWVYEEEMLATKYALWWSPNGKFLAYAEFNDTDIPVIAYSYYG
DEQYPRTINIPYPKAGAKNPVVRIFIIDTTYPAYVGPQEVPVPAMIASSDYYFSWLTWVT
DERVCLQWLKRVQNVSVLSICDFREDWQTWDCPKTQEHIEESRTGWAGGFFVSTPVFSYD
AISYYKIFSDKDGYKHIHYIKDTVENAIQITSGKWEAINIFRVTQDSLFYSSNEFEEYPG
RRNIYRISIGSYPPSKKCVTCHLRKERCQYYTASFSDYAKYYALVCYGPGIPISTLHDGR
TDQEIKILEENKELENALKNIQLPKEEIKKLEVDEITLWYKMILPPQFDRSKKYPLLIQV
YGGPCSQSVRSVFAVNWISYLASKEGMVIALVDGRGTAFQGDKLLYAVYRKLGVYEVEDQ
ITAVRKFIEMGFIDEKRIAIWGWSYGGYVSSLALASGTGLFKCGIAVAPVSSWEYYASVY
TERFMGLPTKDDNLEHYKNSTVMARAEYFRNVDYLLIHGTADDNVHFQNSAQIAKALVNA
QVDFQAMWYSDQNHGLSGLSTNHLYTHMTHFLKQCFSLSD
|
|
|
|---|
| BDBM50338435 |
|---|
| n/a |
|---|
| Name | BDBM50338435 |
|---|
| Synonyms: | (R)-2-(2,5-difluorophenyl)-1-(1-(2-methoxypyrimidin-5-yl)piperidin-4-yl)ethanamine | CHEMBL1683141 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H22F2N4O |
|---|
| Mol. Mass. | 348.3903 |
|---|
| SMILES | COc1ncc(cn1)N1CCC(CC1)[C@H](N)Cc1cc(F)ccc1F |r| |
|---|
| Structure | 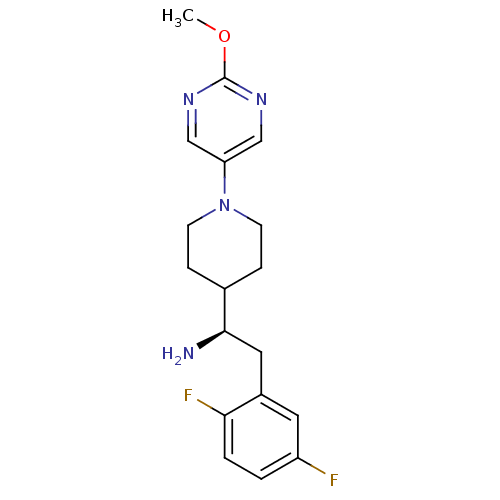
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chen, P; Caldwell, CG; Ashton, W; Wu, JK; He, H; Lyons, KA; Thornberry, NA; Weber, AE Synthesis and evaluation of [(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]cyclohexanes and 4-[(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]piperidines as DPP-4 inhibitors. Bioorg Med Chem Lett21:1880-6 (2011) [PubMed] Article
Chen, P; Caldwell, CG; Ashton, W; Wu, JK; He, H; Lyons, KA; Thornberry, NA; Weber, AE Synthesis and evaluation of [(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]cyclohexanes and 4-[(1R)-1-amino-2-(2,5-difluorophenyl)ethyl]piperidines as DPP-4 inhibitors. Bioorg Med Chem Lett21:1880-6 (2011) [PubMed] Article