| Reaction Details |
|---|
 | Report a problem with these data |
| Target | MAP kinase-activated protein kinase 5 |
|---|
| Ligand | BDBM50341519 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_742151 (CHEMBL1769211) |
|---|
| Ki | >1000±n/a nM |
|---|
| Citation |  Charrier, JD; Miller, A; Kay, DP; Brenchley, G; Twin, HC; Collier, PN; Ramaya, S; Keily, SB; Durrant, SJ; Knegtel, RM; Tanner, AJ; Brown, K; Curnock, AP; Jimenez, JM Discovery and structure-activity relationship of 3-aminopyrid-2-ones as potent and selective interleukin-2 inducible T-cell kinase (Itk) inhibitors. J Med Chem54:2341-50 (2011) [PubMed] Article Charrier, JD; Miller, A; Kay, DP; Brenchley, G; Twin, HC; Collier, PN; Ramaya, S; Keily, SB; Durrant, SJ; Knegtel, RM; Tanner, AJ; Brown, K; Curnock, AP; Jimenez, JM Discovery and structure-activity relationship of 3-aminopyrid-2-ones as potent and selective interleukin-2 inducible T-cell kinase (Itk) inhibitors. J Med Chem54:2341-50 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| MAP kinase-activated protein kinase 5 |
|---|
| Name: | MAP kinase-activated protein kinase 5 |
|---|
| Synonyms: | MAP kinase-activated protein kinase 5 (PRAK) | MAPK-Activated Protein Kinase 5 (MK5) | MAPK-activated protein kinase 5 | MAPK5_HUMAN | MAPKAP kinase 5 | MAPKAPK5 | PRAK | p38-regulated/activated protein kinase | p38-regulated/activated protein kinase (PRAK) |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 54229.92 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant MAPKAPK5 was phosphorylated by incubation with active p38alpha before assays. |
|---|
| Residue: | 473 |
|---|
| Sequence: | MSEESDMDKAIKETSILEEYSINWTQKLGAGISGPVRVCVKKSTQERFALKILLDRPKAR
NEVRLHMMCATHPNIVQIIEVFANSVQFPHESSPRARLLIVMEMMEGGELFHRISQHRHF
TEKQASQVTKQIALALRHCHLLNIAHRDLKPENLLFKDNSLDAPVKLCDFGFAKIDQGDL
MTPQFTPYYVAPQVLEAQRRHQKEKSGIIPTSPTPYTYNKSCDLWSLGVIIYVMLCGYPP
FYSKHHSRTIPKDMRRKIMTGSFEFPEEEWSQISEMAKDVVRKLLKVKPEERLTIEGVLD
HPWLNSTEALDNVLPSAQLMMDKAVVAGIQQAHAEQLANMRIQDLKVSLKPLHSVNNPIL
RKRKLLGTKPKDSVYIHDHENGAEDSNVALEKLRDVIAQCILPQAGKGENEDEKLNEVMQ
EAWKYNRECKLLRDTLQSFSWNGRGFTDKVDRLKLAEIVKQVIEEQTTSHESQ
|
|
|
|---|
| BDBM50341519 |
|---|
| n/a |
|---|
| Name | BDBM50341519 |
|---|
| Synonyms: | (S)-3-(4-(2-(pyrrolidin-1-ylmethyl)pyrrolidin-1-yl)benzoylamino)-5-(6-(methylamino)pyrazin-2-yl)-(1H)-pyridin-2-one | CHEMBL1765781 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H31N7O2 |
|---|
| Mol. Mass. | 473.57 |
|---|
| SMILES | CNc1cncc(n1)-c1c[nH]c(=O)c(NC(=O)c2ccc(cc2)N2CCC[C@H]2CN2CCCC2)c1 |r| |
|---|
| Structure | 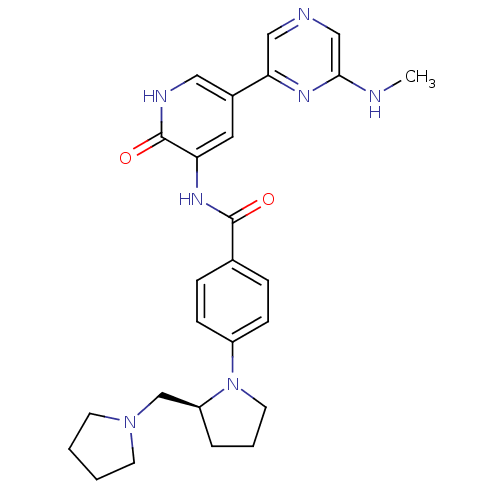
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Charrier, JD; Miller, A; Kay, DP; Brenchley, G; Twin, HC; Collier, PN; Ramaya, S; Keily, SB; Durrant, SJ; Knegtel, RM; Tanner, AJ; Brown, K; Curnock, AP; Jimenez, JM Discovery and structure-activity relationship of 3-aminopyrid-2-ones as potent and selective interleukin-2 inducible T-cell kinase (Itk) inhibitors. J Med Chem54:2341-50 (2011) [PubMed] Article
Charrier, JD; Miller, A; Kay, DP; Brenchley, G; Twin, HC; Collier, PN; Ramaya, S; Keily, SB; Durrant, SJ; Knegtel, RM; Tanner, AJ; Brown, K; Curnock, AP; Jimenez, JM Discovery and structure-activity relationship of 3-aminopyrid-2-ones as potent and selective interleukin-2 inducible T-cell kinase (Itk) inhibitors. J Med Chem54:2341-50 (2011) [PubMed] Article