| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nitric oxide synthase, brain |
|---|
| Ligand | BDBM50330882 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_743323 (CHEMBL1767570) |
|---|
| Ki | 5.3±n/a nM |
|---|
| Citation |  Xue, F; Fang, J; Delker, SL; Li, H; Martásek, P; Roman, LJ; Poulos, TL; Silverman, RB Symmetric double-headed aminopyridines, a novel strategy for potent and membrane-permeable inhibitors of neuronal nitric oxide synthase. J Med Chem54:2039-48 (2011) [PubMed] Article Xue, F; Fang, J; Delker, SL; Li, H; Martásek, P; Roman, LJ; Poulos, TL; Silverman, RB Symmetric double-headed aminopyridines, a novel strategy for potent and membrane-permeable inhibitors of neuronal nitric oxide synthase. J Med Chem54:2039-48 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Nitric oxide synthase, brain |
|---|
| Name: | Nitric oxide synthase, brain |
|---|
| Synonyms: | Constitutive NOS | N-NOS | NC-NOS | NOS type I | NOS type I nNOS | NOS1 | NOS1_HUMAN | Neuronal NOS | Neuronal nitric oxide synthase | Nitric oxide synthase, brain (nNOS) | Nitric oxide synthase, neuronal (nNOS) | Peptidyl-cysteine S-nitrosylase NOS1 | bNOS | nNOS |
|---|
| Type: | Homodimer |
|---|
| Mol. Mass.: | 160985.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29475 |
|---|
| Residue: | 1434 |
|---|
| Sequence: | MEDHMFGVQQIQPNVISVRLFKRKVGGLGFLVKERVSKPPVIISDLIRGGAAEQSGLIQA
GDIILAVNGRPLVDLSYDSALEVLRGIASETHVVLILRGPEGFTTHLETTFTGDGTPKTI
RVTQPLGPPTKAVDLSHQPPAGKEQPLAVDGASGPGNGPQHAYDDGQEAGSLPHANGLAP
RPPGQDPAKKATRVSLQGRGENNELLKEIEPVLSLLTSGSRGVKGGAPAKAEMKDMGIQV
DRDLDGKSHKPLPLGVENDRVFNDLWGKGNVPVVLNNPYSEKEQPPTSGKQSPTKNGSPS
KCPRFLKVKNWETEVVLTDTLHLKSTLETGCTEYICMGSIMHPSQHARRPEDVRTKGQLF
PLAKEFIDQYYSSIKRFGSKAHMERLEEVNKEIDTTSTYQLKDTELIYGAKHAWRNASRC
VGRIQWSKLQVFDARDCTTAHGMFNYICNHVKYATNKGNLRSAITIFPQRTDGKHDFRVW
NSQLIRYAGYKQPDGSTLGDPANVQFTEICIQQGWKPPRGRFDVLPLLLQANGNDPELFQ
IPPELVLEVPIRHPKFEWFKDLGLKWYGLPAVSNMLLEIGGLEFSACPFSGWYMGTEIGV
RDYCDNSRYNILEEVAKKMNLDMRKTSSLWKDQALVEINIAVLYSFQSDKVTIVDHHSAT
ESFIKHMENEYRCRGGCPADWVWIVPPMSGSITPVFHQEMLNYRLTPSFEYQPDPWNTHV
WKGTNGTPTKRRAIGFKKLAEAVKFSAKLMGQAMAKRVKATILYATETGKSQAYAKTLCE
IFKHAFDAKVMSMEEYDIVHLEHETLVLVVTSTFGNGDPPENGEKFGCALMEMRHPNSVQ
EERKSYKVRFNSVSSYSDSQKSSGDGPDLRDNFESAGPLANVRFSVFGLGSRAYPHFCAF
GHAVDTLLEELGGERILKMREGDELCGQEEAFRTWAKKVFKAACDVFCVGDDVNIEKANN
SLISNDRSWKRNKFRLTFVAEAPELTQGLSNVHKKRVSAARLLSRQNLQSPKSSRSTIFV
RLHTNGSQELQYQPGDHLGVFPGNHEDLVNALIERLEDAPPVNQMVKVELLEERNTALGV
ISNWTDELRLPPCTIFQAFKYYLDITTPPTPLQLQQFASLATSEKEKQRLLVLSKGLQEY
EEWKWGKNPTIVEVLEEFPSIQMPATLLLTQLSLLQPRYYSISSSPDMYPDEVHLTVAIV
SYRTRDGEGPIHHGVCSSWLNRIQADELVPCFVRGAPSFHLPRNPQVPCILVGPGTGIAP
FRSFWQQRQFDIQHKGMNPCPMVLVFGCRQSKIDHIYREETLQAKNKGVFRELYTAYSRE
PDKPKKYVQDILQEQLAESVYRALKEQGGHIYVCGDVTMAADVLKAIQRIMTQQGKLSAE
DAGVFISRMRDDNRYHEDIFGVTLRTYEVTNRLRSESIAFIEESKKDTDEVFSS
|
|
|
|---|
| BDBM50330882 |
|---|
| n/a |
|---|
| Name | BDBM50330882 |
|---|
| Synonyms: | CHEMBL1277951 | CHEMBL594682 | N1-((3R,4R)-4-((6-aminopyridin-2-yl)methyl)pyrrolidin-3-yl)-N2-(4-fluorobenzyl)ethane-1,2-diamine tetrahydrochloride | US9090589, 1 | US9212144, 1 | US9783500, Compound 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H30FN5 |
|---|
| Mol. Mass. | 371.4948 |
|---|
| SMILES | Cc1cc(N)nc(C[C@@H]2CNC[C@@H]2NCCNCCc2cccc(F)c2)c1 |r| |
|---|
| Structure | 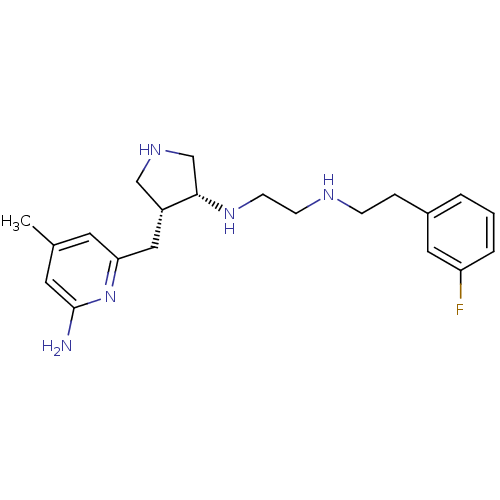
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xue, F; Fang, J; Delker, SL; Li, H; Martásek, P; Roman, LJ; Poulos, TL; Silverman, RB Symmetric double-headed aminopyridines, a novel strategy for potent and membrane-permeable inhibitors of neuronal nitric oxide synthase. J Med Chem54:2039-48 (2011) [PubMed] Article
Xue, F; Fang, J; Delker, SL; Li, H; Martásek, P; Roman, LJ; Poulos, TL; Silverman, RB Symmetric double-headed aminopyridines, a novel strategy for potent and membrane-permeable inhibitors of neuronal nitric oxide synthase. J Med Chem54:2039-48 (2011) [PubMed] Article