| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50319981 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_771460 (CHEMBL1837220) |
|---|
| IC50 | 2600±n/a nM |
|---|
| Citation |  Mohamed, T; Yeung, JC; Rao, PP Development of 2-substituted-N-(naphth-1-ylmethyl) and N-benzhydrylpyrimidin-4-amines as dual cholinesterase and Aß-aggregation inhibitors: Synthesis and biological evaluation. Bioorg Med Chem Lett21:5881-7 (2011) [PubMed] Article Mohamed, T; Yeung, JC; Rao, PP Development of 2-substituted-N-(naphth-1-ylmethyl) and N-benzhydrylpyrimidin-4-amines as dual cholinesterase and Aß-aggregation inhibitors: Synthesis and biological evaluation. Bioorg Med Chem Lett21:5881-7 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | BCHE | Butyrylcholinesterase (BuChE) | CHLE_HORSE | Cholinesterase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65643.35 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | P81908 |
|---|
| Residue: | 574 |
|---|
| Sequence: | EEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATK
YANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQT
GTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQK
NIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEAR
NRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLT
DMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPR
VSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTRKFSELGNDAFFYY
FEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTRAEEILSRSIMKRWANFAKYGNP
NGTQNNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAE
REWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
|
|
|
|---|
| BDBM50319981 |
|---|
| n/a |
|---|
| Name | BDBM50319981 |
|---|
| Synonyms: | 2-(4-methylpiperazin-1-yl)-N-(naphthalen-1-ylmethyl)pyrimidin-4-amine | CHEMBL1084213 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H23N5 |
|---|
| Mol. Mass. | 333.4301 |
|---|
| SMILES | CN1CCN(CC1)c1nccc(NCc2cccc3ccccc23)n1 |
|---|
| Structure | 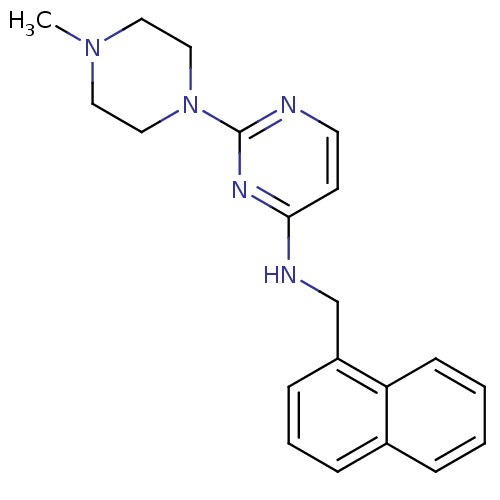
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mohamed, T; Yeung, JC; Rao, PP Development of 2-substituted-N-(naphth-1-ylmethyl) and N-benzhydrylpyrimidin-4-amines as dual cholinesterase and Aß-aggregation inhibitors: Synthesis and biological evaluation. Bioorg Med Chem Lett21:5881-7 (2011) [PubMed] Article
Mohamed, T; Yeung, JC; Rao, PP Development of 2-substituted-N-(naphth-1-ylmethyl) and N-benzhydrylpyrimidin-4-amines as dual cholinesterase and Aß-aggregation inhibitors: Synthesis and biological evaluation. Bioorg Med Chem Lett21:5881-7 (2011) [PubMed] Article