| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50293721 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_788861 (CHEMBL1924931) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Jones, CK; Engers, DW; Thompson, AD; Field, JR; Blobaum, AL; Lindsley, SR; Zhou, Y; Gogliotti, RD; Jadhav, S; Zamorano, R; Bogenpohl, J; Smith, Y; Morrison, R; Daniels, JS; Weaver, CD; Conn, PJ; Lindsley, CW; Niswender, CM; Hopkins, CR Discovery, synthesis, and structure-activity relationship development of a series of N-4-(2,5-dioxopyrrolidin-1-yl)phenylpicolinamides (VU0400195, ML182): characterization of a novel positive allosteric modulator of the metabotropic glutamate receptor 4 (mGlu(4)) with oral efficacy in an antiparkin J Med Chem54:7639-47 (2011) [PubMed] Article Jones, CK; Engers, DW; Thompson, AD; Field, JR; Blobaum, AL; Lindsley, SR; Zhou, Y; Gogliotti, RD; Jadhav, S; Zamorano, R; Bogenpohl, J; Smith, Y; Morrison, R; Daniels, JS; Weaver, CD; Conn, PJ; Lindsley, CW; Niswender, CM; Hopkins, CR Discovery, synthesis, and structure-activity relationship development of a series of N-4-(2,5-dioxopyrrolidin-1-yl)phenylpicolinamides (VU0400195, ML182): characterization of a novel positive allosteric modulator of the metabotropic glutamate receptor 4 (mGlu(4)) with oral efficacy in an antiparkin J Med Chem54:7639-47 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50293721 |
|---|
| n/a |
|---|
| Name | BDBM50293721 |
|---|
| Synonyms: | CHEMBL562551 | N-(4-chloro-3-methoxyphenyl)picolinamide | cid_44191096 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H11ClN2O2 |
|---|
| Mol. Mass. | 262.692 |
|---|
| SMILES | COc1cc(NC(=O)c2ccccn2)ccc1Cl |
|---|
| Structure | 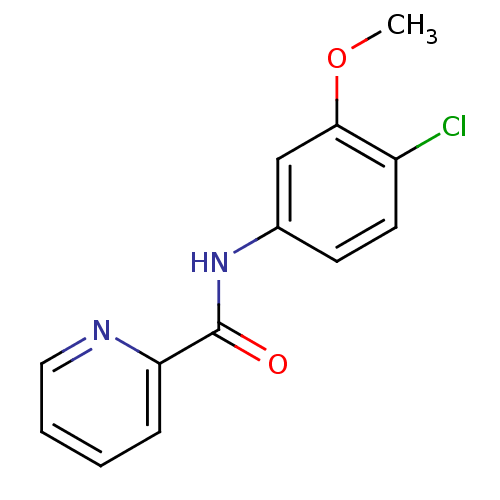
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jones, CK; Engers, DW; Thompson, AD; Field, JR; Blobaum, AL; Lindsley, SR; Zhou, Y; Gogliotti, RD; Jadhav, S; Zamorano, R; Bogenpohl, J; Smith, Y; Morrison, R; Daniels, JS; Weaver, CD; Conn, PJ; Lindsley, CW; Niswender, CM; Hopkins, CR Discovery, synthesis, and structure-activity relationship development of a series of N-4-(2,5-dioxopyrrolidin-1-yl)phenylpicolinamides (VU0400195, ML182): characterization of a novel positive allosteric modulator of the metabotropic glutamate receptor 4 (mGlu(4)) with oral efficacy in an antiparkin J Med Chem54:7639-47 (2011) [PubMed] Article
Jones, CK; Engers, DW; Thompson, AD; Field, JR; Blobaum, AL; Lindsley, SR; Zhou, Y; Gogliotti, RD; Jadhav, S; Zamorano, R; Bogenpohl, J; Smith, Y; Morrison, R; Daniels, JS; Weaver, CD; Conn, PJ; Lindsley, CW; Niswender, CM; Hopkins, CR Discovery, synthesis, and structure-activity relationship development of a series of N-4-(2,5-dioxopyrrolidin-1-yl)phenylpicolinamides (VU0400195, ML182): characterization of a novel positive allosteric modulator of the metabotropic glutamate receptor 4 (mGlu(4)) with oral efficacy in an antiparkin J Med Chem54:7639-47 (2011) [PubMed] Article