| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lysosomal acid glucosylceramidase |
|---|
| Ligand | BDBM50283602 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_37555 (CHEMBL647636) |
|---|
| IC50 | 4890±n/a nM |
|---|
| Citation |  Uchida, C; Kimura, H; Ogawa, S Potent glycosidase inhibitors, N-phenyl cyclic isourea derivatives of 5-amino- and 5-amino-1-C-(hydroxymethyl)-cyclopentane-1,2,3,4-tetraols Bioorg Med Chem Lett4:2643-2648 (1994) Article Uchida, C; Kimura, H; Ogawa, S Potent glycosidase inhibitors, N-phenyl cyclic isourea derivatives of 5-amino- and 5-amino-1-C-(hydroxymethyl)-cyclopentane-1,2,3,4-tetraols Bioorg Med Chem Lett4:2643-2648 (1994) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lysosomal acid glucosylceramidase |
|---|
| Name: | Lysosomal acid glucosylceramidase |
|---|
| Synonyms: | Acid beta-glucosidase | Alglucerase | Beta-glucocerebrosidase | Beta-glucocerebrosidase (GC) | D-glucosyl-N-acylsphingosine glucohydrolase | GBA | GBA1 | GBA1_HUMAN | GC | GCase | GLUC | Glucocerebrosidase (GBA) | Glucosylceramidase (GBA) | Glucosylceramidase (GCase) | Glucosylceramidase precursor (Beta-glucocerebrosidase) (Acid beta-glucosidase) (D-glucosyl-N-acylsphingosine glucohydrolase) (Alglucerase) (Imiglucerase) | Imiglucerase | beta-glucocerebrosidase (GCase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 59724.64 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The beta-Glu activity was measured with commercially available beta-glucocerebrosidase (Ceredase) as the enzyme source. |
|---|
| Residue: | 536 |
|---|
| Sequence: | MEFSSPSREECPKPLSRVSIMAGSLTGLLLLQAVSWASGARPCIPKSFGYSSVVCVCNAT
YCDSFDPPTFPALGTFSRYESTRSGRRMELSMGPIQANHTGTGLLLTLQPEQKFQKVKGF
GGAMTDAAALNILALSPPAQNLLLKSYFSEEGIGYNIIRVPMASCDFSIRTYTYADTPDD
FQLHNFSLPEEDTKLKIPLIHRALQLAQRPVSLLASPWTSPTWLKTNGAVNGKGSLKGQP
GDIYHQTWARYFVKFLDAYAEHKLQFWAVTAENEPSAGLLSGYPFQCLGFTPEHQRDFIA
RDLGPTLANSTHHNVRLLMLDDQRLLLPHWAKVVLTDPEAAKYVHGIAVHWYLDFLAPAK
ATLGETHRLFPNTMLFASEACVGSKFWEQSVRLGSWDRGMQYSHSIITNLLYHVVGWTDW
NLALNPEGGPNWVRNFVDSPIIVDITKDTFYKQPMFYHLGHFSKFIPEGSQRVGLVASQK
NDLDAVALMHPDGSAVVVVLNRSSKDVPLTIKDPAVGFLETISPGYSIHTYLWRRQ
|
|
|
|---|
| BDBM50283602 |
|---|
| n/a |
|---|
| Name | BDBM50283602 |
|---|
| Synonyms: | 6a-Hydroxymethyl-2-phenylamino-4,5,6,6a-tetrahydro-3aH-cyclopentaoxazole-4,5,6-triol | CHEMBL86772 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H16N2O5 |
|---|
| Mol. Mass. | 280.2765 |
|---|
| SMILES | OCC12OC(Nc3ccccc3)=NC1C(O)C(O)C2O |c:12| |
|---|
| Structure | 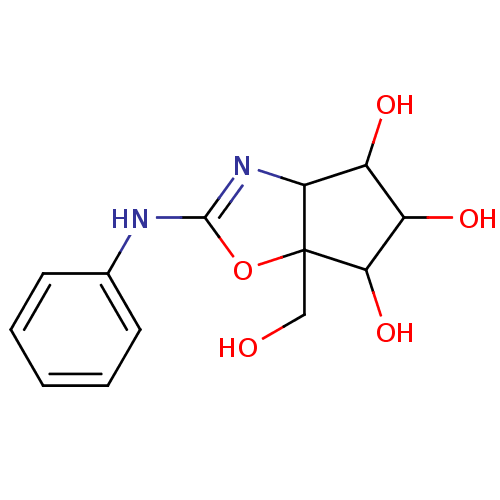
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Uchida, C; Kimura, H; Ogawa, S Potent glycosidase inhibitors, N-phenyl cyclic isourea derivatives of 5-amino- and 5-amino-1-C-(hydroxymethyl)-cyclopentane-1,2,3,4-tetraols Bioorg Med Chem Lett4:2643-2648 (1994) Article
Uchida, C; Kimura, H; Ogawa, S Potent glycosidase inhibitors, N-phenyl cyclic isourea derivatives of 5-amino- and 5-amino-1-C-(hydroxymethyl)-cyclopentane-1,2,3,4-tetraols Bioorg Med Chem Lett4:2643-2648 (1994) Article