| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Transient receptor potential cation channel subfamily V member 1 |
|---|
| Ligand | BDBM50366620 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_215635 (CHEMBL820953) |
|---|
| Ki | 0.120±n/a nM |
|---|
| Citation |  Walpole, CS; Bevan, S; Bloomfield, G; Breckenridge, R; James, IF; Ritchie, T; Szallasi, A; Winter, J; Wrigglesworth, R Similarities and differences in the structure-activity relationships of capsaicin and resiniferatoxin analogues. J Med Chem39:2939-52 (1996) [PubMed] Article Walpole, CS; Bevan, S; Bloomfield, G; Breckenridge, R; James, IF; Ritchie, T; Szallasi, A; Winter, J; Wrigglesworth, R Similarities and differences in the structure-activity relationships of capsaicin and resiniferatoxin analogues. J Med Chem39:2939-52 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Transient receptor potential cation channel subfamily V member 1 |
|---|
| Name: | Transient receptor potential cation channel subfamily V member 1 |
|---|
| Synonyms: | Capsaicin receptor | OTRPC1 | TRPV1_RAT | Transient receptor potential cation channel subfamily V member 1 (TRPV1) | Trpv1 | Vanilloid Receptor 1 (TRPV1, VR1) | Vanilloid VR1 | Vanilloid receptor | Vanilloid receptor 1 (VRI/TRPV1) | Vanilloid receptor type 1-like | Vanilloid receptor type 1-like (TrpV1/Vr1) | Vr1 | Vr1l | osm-9-like TRP channel 1 |
|---|
| Type: | Transient Receptor |
|---|
| Mol. Mass.: | 94956.12 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | O35433 |
|---|
| Residue: | 838 |
|---|
| Sequence: | MEQRASLDSEESESPPQENSCLDPPDRDPNCKPPPVKPHIFTTRSRTRLFGKGDSEEASP
LDCPYEEGGLASCPIITVSSVLTIQRPGDGPASVRPSSQDSVSAGEKPPRLYDRRSIFDA
VAQSNCQELESLLPFLQRSKKRLTDSEFKDPETGKTCLLKAMLNLHNGQNDTIALLLDVA
RKTDSLKQFVNASYTDSYYKGQTALHIAIERRNMTLVTLLVENGADVQAAANGDFFKKTK
GRPGFYFGELPLSLAACTNQLAIVKFLLQNSWQPADISARDSVGNTVLHALVEVADNTVD
NTKFVTSMYNEILILGAKLHPTLKLEEITNRKGLTPLALAASSGKIGVLAYILQREIHEP
ECRHLSRKFTEWAYGPVHSSLYDLSCIDTCEKNSVLEVIAYSSSETPNRHDMLLVEPLNR
LLQDKWDRFVKRIFYFNFFVYCLYMIIFTAAAYYRPVEGLPPYKLKNTVGDYFRVTGEIL
SVSGGVYFFFRGIQYFLQRRPSLKSLFVDSYSEILFFVQSLFMLVSVVLYFSQRKEYVAS
MVFSLAMGWTNMLYYTRGFQQMGIYAVMIEKMILRDLCRFMFVYLVFLFGFSTAVVTLIE
DGKNNSLPMESTPHKCRGSACKPGNSYNSLYSTCLELFKFTIGMGDLEFTENYDFKAVFI
ILLLAYVILTYILLLNMLIALMGETVNKIAQESKNIWKLQRAITILDTEKSFLKCMRKAF
RSGKLLQVGFTPDGKDDYRWCFRVDEVNWTTWNTNVGIINEDPGNCEGVKRTLSFSLRSG
RVSGRNWKNFALVPLLRDASTRDRHATQQEEVQLKHYTGSLKPEDAEVFKDSMVPGEK
|
|
|
|---|
| BDBM50366620 |
|---|
| n/a |
|---|
| Name | BDBM50366620 |
|---|
| Synonyms: | RESINIFERATOXIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H40O9 |
|---|
| Mol. Mass. | 628.7081 |
|---|
| SMILES | COc1cc(CC(=O)OCC2=C[C@H]3[C@H]4OC5(Cc6ccccc6)O[C@@]4(C[C@@H](C)[C@]3(O5)[C@@H]3C=C(C)C(=O)[C@@]3(O)C2)C(C)=C)ccc1O |r,t:10,35,TLB:23:15:12:24.25.26,THB:16:15:12:24.25.26| |
|---|
| Structure | 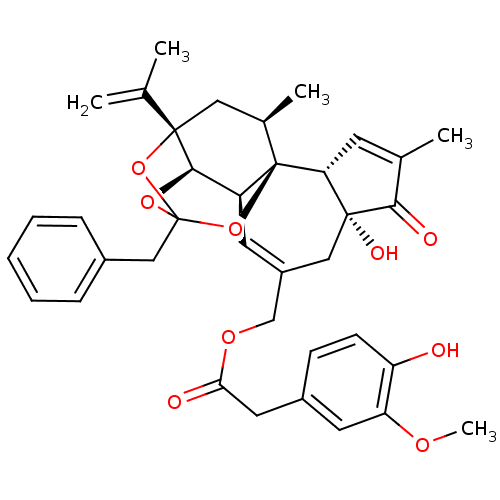
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Walpole, CS; Bevan, S; Bloomfield, G; Breckenridge, R; James, IF; Ritchie, T; Szallasi, A; Winter, J; Wrigglesworth, R Similarities and differences in the structure-activity relationships of capsaicin and resiniferatoxin analogues. J Med Chem39:2939-52 (1996) [PubMed] Article
Walpole, CS; Bevan, S; Bloomfield, G; Breckenridge, R; James, IF; Ritchie, T; Szallasi, A; Winter, J; Wrigglesworth, R Similarities and differences in the structure-activity relationships of capsaicin and resiniferatoxin analogues. J Med Chem39:2939-52 (1996) [PubMed] Article