| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Progesterone receptor |
|---|
| Ligand | BDBM50148732 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_159052 (CHEMBL760814) |
|---|
| EC50 | 2.2±n/a nM |
|---|
| Citation |  Edwards, JP; West, SJ; Marschke, KB; Mais, DE; Gottardis, MM; Jones, TK 5-Aryl-1,2-dihydro-5H-chromeno[3,4-f]quinolines as potent, orally active, nonsteroidal progesterone receptor agonists: the effect of D-ring substituents. J Med Chem41:303-10 (1998) [PubMed] Article Edwards, JP; West, SJ; Marschke, KB; Mais, DE; Gottardis, MM; Jones, TK 5-Aryl-1,2-dihydro-5H-chromeno[3,4-f]quinolines as potent, orally active, nonsteroidal progesterone receptor agonists: the effect of D-ring substituents. J Med Chem41:303-10 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Progesterone receptor |
|---|
| Name: | Progesterone receptor |
|---|
| Synonyms: | NR3C3 | Nuclear receptor subfamily 3 group C member 3 | PGR | PR | PRGR_HUMAN | progesterone |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 98979.52 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06401 |
|---|
| Residue: | 933 |
|---|
| Sequence: | MTELKAKGPRAPHVAGGPPSPEVGSPLLCRPAAGPFPGSQTSDTLPEVSAIPISLDGLLF
PRPCQGQDPSDEKTQDQQSLSDVEGAYSRAEATRGAGGSSSSPPEKDSGLLDSVLDTLLA
PSGPGQSQPSPPACEVTSSWCLFGPELPEDPPAAPATQRVLSPLMSRSGCKVGDSSGTAA
AHKVLPRGLSPARQLLLPASESPHWSGAPVKPSPQAAAVEVEEEDGSESEESAGPLLKGK
PRALGGAAAGGGAAAVPPGAAAGGVALVPKEDSRFSAPRVALVEQDAPMAPGRSPLATTV
MDFIHVPILPLNHALLAARTRQLLEDESYDGGAGAASAFAPPRSSPCASSTPVAVGDFPD
CAYPPDAEPKDDAYPLYSDFQPPALKIKEEEEGAEASARSPRSYLVAGANPAAFPDFPLG
PPPPLPPRATPSRPGEAAVTAAPASASVSSASSSGSTLECILYKAEGAPPQQGPFAPPPC
KAPGASGCLLPRDGLPSTSASAAAAGAAPALYPALGLNGLPQLGYQAAVLKEGLPQVYPP
YLNYLRPDSEASQSPQYSFESLPQKICLICGDEASGCHYGVLTCGSCKVFFKRAMEGQHN
YLCAGRNDCIVDKIRRKNCPACRLRKCCQAGMVLGGRKFKKFNKVRVVRALDAVALPQPV
GVPNESQALSQRFTFSPGQDIQLIPPLINLLMSIEPDVIYAGHDNTKPDTSSSLLTSLNQ
LGERQLLSVVKWSKSLPGFRNLHIDDQITLIQYSWMSLMVFGLGWRSYKHVSGQMLYFAP
DLILNEQRMKESSFYSLCLTMWQIPQEFVKLQVSQEEFLCMKVLLLLNTIPLEGLRSQTQ
FEEMRSSYIRELIKAIGLRQKGVVSSSQRFYQLTKLLDNLHDLVKQLHLYCLNTFIQSRA
LSVEFPEMMSEVIAAQLPKILAGMVKPLLFHKK
|
|
|
|---|
| BDBM50148732 |
|---|
| n/a |
|---|
| Name | BDBM50148732 |
|---|
| Synonyms: | 17-ethynyl-17beta-hydroxyestr-4-en-3-one | 17alpha-ethinyl-19-nortestosterone | 17alpha-ethinylestra-4-en-17beta-ol-3-one | 17alpha-ethynyl-19-nor-4-androsten-17beta-ol-3-one | 17beta-hydroxy-19-norpregn-4-en-20-yn-3-one | 19-Nor-17alpha-ethynyl-17beta-hydroxy-4-androsten-3-one | 19-Norethisterone | 19-nor-17alpha-ethynyltestosterone | 4-estren-17alpha-ethynyl-17beta-ol-3-one | CHEMBL1162 | Micronor | NORETHINDRONE | Norethisteron | Primolut-N | norethisterone |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H26O2 |
|---|
| Mol. Mass. | 298.4192 |
|---|
| SMILES | C[C@]12CC[C@H]3[C@@H](CCC4=CC(=O)CC[C@H]34)[C@@H]1CC[C@@]2(O)C#C |t:8| |
|---|
| Structure | 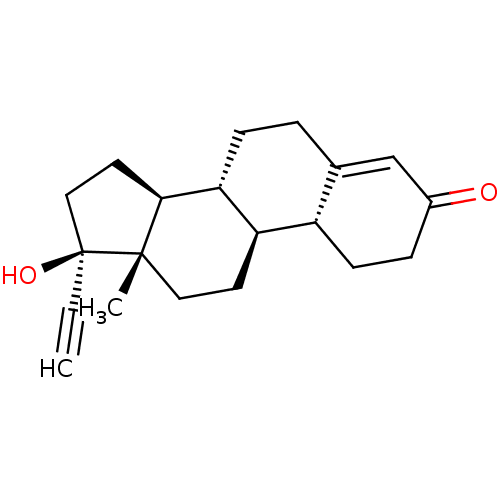
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Edwards, JP; West, SJ; Marschke, KB; Mais, DE; Gottardis, MM; Jones, TK 5-Aryl-1,2-dihydro-5H-chromeno[3,4-f]quinolines as potent, orally active, nonsteroidal progesterone receptor agonists: the effect of D-ring substituents. J Med Chem41:303-10 (1998) [PubMed] Article
Edwards, JP; West, SJ; Marschke, KB; Mais, DE; Gottardis, MM; Jones, TK 5-Aryl-1,2-dihydro-5H-chromeno[3,4-f]quinolines as potent, orally active, nonsteroidal progesterone receptor agonists: the effect of D-ring substituents. J Med Chem41:303-10 (1998) [PubMed] Article