| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Ligand | BDBM50192451 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_209306 (CHEMBL811761) |
|---|
| Ki | 4.7±n/a nM |
|---|
| Citation |  McKie, JH; Douglas, KT; Chan, C; Roser, SA; Yates, R; Read, M; Hyde, JE; Dascombe, MJ; Yuthavong, Y; Sirawaraporn, W Rational drug design approach for overcoming drug resistance: application to pyrimethamine resistance in malaria. J Med Chem41:1367-70 (1998) [PubMed] Article McKie, JH; Douglas, KT; Chan, C; Roser, SA; Yates, R; Read, M; Hyde, JE; Dascombe, MJ; Yuthavong, Y; Sirawaraporn, W Rational drug design approach for overcoming drug resistance: application to pyrimethamine resistance in malaria. J Med Chem41:1367-70 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Name: | Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Synonyms: | DHFR-TS | DRTS_PLAFK | Dihydrofolate Reductase-Thymidylate Synthase (DHFR-TS) Mutant KICB1 | Dihydrofolate reductase | PfDHFR-TS double mutant (C59R+S108N) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 71822.51 |
|---|
| Organism: | Plasmodium falciparum (isolate K1 / Thailand) |
|---|
| Description: | The mutant clone was prepared by cassette mutagenesis using wildtype pfDHFR as a template, and expressed in E. coli. |
|---|
| Residue: | 608 |
|---|
| Sequence: | MMEQVCDVFDIYAICACCKVESKNEGKKNEVFNNYTFRGLGNKGVLPWKCNSLDMKYFRA
VTTYVNESKYEKLKYKRCKYLNKETVDNVNDMPNSKKLQNVVVMGRTNWESIPKKFKPLS
NRINVILSRTLKKEDFDEDVYIINKVEDLIVLLGKLNYYKCFIIGGSVVYQEFLEKKLIK
KIYFTRINSTYECDVFFPEINENEYQIISVSDVYTSNNTTLDFIIYKKTNNKMLNEQNCI
KGEEKNNDMPLKNDDKDTCHMKKLTEFYKNVDKYKINYENDDDDEEEDDFVYFNFNKEKE
EKNKNSIHPNDFQIYNSLKYKYHPEYQYLNIIYDIMMNGNKQSDRTGVGVLSKFGYIMKF
DLSQYFPLLTTKKLFLRGIIEELLWFIRGETNGNTLLNKNVRIWEANGTREFLDNRKLFH
REVNDLGPIYGFQWRHFGAEYTNMYDNYENKGVDQLKNIINLIKNDPTSRRILLCAWNVK
DLDQMALPPCHILCQFYVFDGKLSCIMYQRSCDLGLGVPFNIASYSIFTHMIAQVCNLQP
AQFIHVLGNAHVYNNHIDSLKIQLNRIPYPFPTLKLNPDIKNIEDFTISDFTIQNYVHHE
KISMDMAA
|
|
|
|---|
| BDBM50192451 |
|---|
| n/a |
|---|
| Name | BDBM50192451 |
|---|
| Synonyms: | CHEMBL213053 | NADP+ |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H25N7O17P3 |
|---|
| Mol. Mass. | 740.3828 |
|---|
| SMILES | NC(=O)c1ccc[n+](c1)C1OC(COP([O-])(=O)OP([O-])(=O)OC[C@H]2O[C@H]([C@H](OP([O-])([O-])=O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@H](O)[C@@H]1O |
|---|
| Structure | 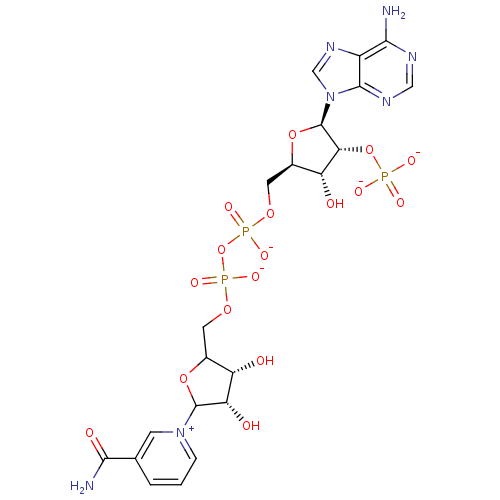
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 McKie, JH; Douglas, KT; Chan, C; Roser, SA; Yates, R; Read, M; Hyde, JE; Dascombe, MJ; Yuthavong, Y; Sirawaraporn, W Rational drug design approach for overcoming drug resistance: application to pyrimethamine resistance in malaria. J Med Chem41:1367-70 (1998) [PubMed] Article
McKie, JH; Douglas, KT; Chan, C; Roser, SA; Yates, R; Read, M; Hyde, JE; Dascombe, MJ; Yuthavong, Y; Sirawaraporn, W Rational drug design approach for overcoming drug resistance: application to pyrimethamine resistance in malaria. J Med Chem41:1367-70 (1998) [PubMed] Article