| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Kappa-type opioid receptor |
|---|
| Ligand | BDBM50066535 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_145381 (CHEMBL750071) |
|---|
| Ki | 1.60±n/a nM |
|---|
| Citation |  Derrick, I; Neilan, CL; Andes, J; Husbands, SM; Woods, JH; Traynor, JR; Lewis, JW 3-Deoxyclocinnamox: the first high-affinity, nonpeptide mu-opioid antagonist lacking a phenolic hydroxyl group. J Med Chem43:3348-50 (2000) [PubMed] Derrick, I; Neilan, CL; Andes, J; Husbands, SM; Woods, JH; Traynor, JR; Lewis, JW 3-Deoxyclocinnamox: the first high-affinity, nonpeptide mu-opioid antagonist lacking a phenolic hydroxyl group. J Med Chem43:3348-50 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Kappa-type opioid receptor |
|---|
| Name: | Kappa-type opioid receptor |
|---|
| Synonyms: | K-OR-1 | KOR-1 | Kappa-opioid receptor (KOR) | Kappa-type opioid receptor (KOPR) | Kappa-type opioid receptor (KOR) | Kappa-type opioid receptor (Kappa) | OPIATE Kappa | OPRK | OPRK1 | OPRK_HUMAN | kappa opioid receptor (KOR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 42648.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P41145 |
|---|
| Residue: | 380 |
|---|
| Sequence: | MDSPIQIFRGEPGPTCAPSACLPPNSSAWFPGWAEPDSNGSAGSEDAQLEPAHISPAIPV
IITAVYSVVFVVGLVGNSLVMFVIIRYTKMKTATNIYIFNLALADALVTTTMPFQSTVYL
MNSWPFGDVLCKIVISIDYYNMFTSIFTLTMMSVDRYIAVCHPVKALDFRTPLKAKIINI
CIWLLSSSVGISAIVLGGTKVREDVDVIECSLQFPDDDYSWWDLFMKICVFIFAFVIPVL
IIIVCYTLMILRLKSVRLLSGSREKDRNLRRITRLVLVVVAVFVVCWTPIHIFILVEALG
STSHSTAALSSYYFCIALGYTNSSLNPILYAFLDENFKRCFRDFCFPLKMRMERQSTSRV
RNTVQDPAYLRDIDGMNKPV
|
|
|
|---|
| BDBM50066535 |
|---|
| n/a |
|---|
| Name | BDBM50066535 |
|---|
| Synonyms: | (2E)-3-(4-chlorophenyl)-N-[(1R,13R,17S)-4-(cyclopropylmethyl)-10-hydroxy-14-oxo-12-oxa-4-azapentacyclo[9.6.1.0^{1,13}.0^{5,17}.0^{7,18}]octadeca-7,9,11(18)-trien-17-yl]prop-2-enamide | 14Beta-4'-Chlorocinnamoylaminodihydronormorphinone | 1N-[4-cyclopropylmethyl-10-hydroxy-14-oxo-(17S)-12-oxa-4-azapentacyclo[9.6.1.01,13.05,17.07,18]octadeca-7,9,11(18)-trien-17-yl]-3-(4-chlorophenyl)-(E)-2-propenamide(Clocinnamox (C-CAM)) | Clocinnamox |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H29ClN2O4 |
|---|
| Mol. Mass. | 505.005 |
|---|
| SMILES | [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC6CC6)CC[C@@]14[C@]5(CCC2=O)NC(=O)\C=C\c1ccc(Cl)cc1)ccc3O |r,THB:10:9:17:6.5.4| |
|---|
| Structure | 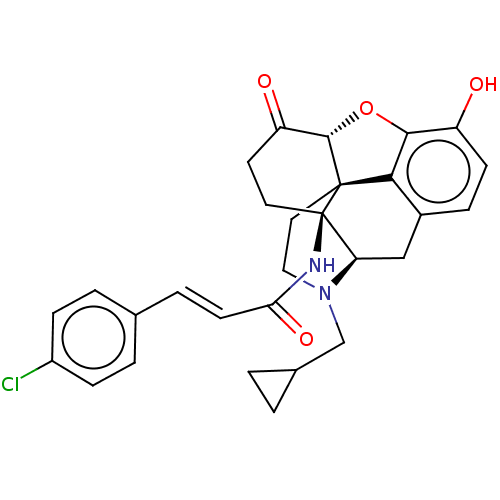
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Derrick, I; Neilan, CL; Andes, J; Husbands, SM; Woods, JH; Traynor, JR; Lewis, JW 3-Deoxyclocinnamox: the first high-affinity, nonpeptide mu-opioid antagonist lacking a phenolic hydroxyl group. J Med Chem43:3348-50 (2000) [PubMed]
Derrick, I; Neilan, CL; Andes, J; Husbands, SM; Woods, JH; Traynor, JR; Lewis, JW 3-Deoxyclocinnamox: the first high-affinity, nonpeptide mu-opioid antagonist lacking a phenolic hydroxyl group. J Med Chem43:3348-50 (2000) [PubMed]