| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin D |
|---|
| Ligand | BDBM7974 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_45182 (CHEMBL658681) |
|---|
| IC50 | 21±n/a nM |
|---|
| Citation |  Leung, D; Abbenante, G; Fairlie, DP Protease inhibitors: current status and future prospects. J Med Chem43:305-41 (2000) [PubMed] Leung, D; Abbenante, G; Fairlie, DP Protease inhibitors: current status and future prospects. J Med Chem43:305-41 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin D |
|---|
| Name: | Cathepsin D |
|---|
| Synonyms: | CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44551.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated. |
|---|
| Residue: | 412 |
|---|
| Sequence: | MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVP
AVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIH
HKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFG
EATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQ
PGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSL
MVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQ
AGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
|
|
|
|---|
| BDBM7974 |
|---|
| n/a |
|---|
| Name | BDBM7974 |
|---|
| Synonyms: | (3S,4S)-N-[(1S)-1-{[(1S)-1-carbamoyl-3-methylbutyl]carbamoyl}ethyl]-3-hydroxy-4-[(2S)-3-methyl-2-(pyridin-2-ylformamido)butanamido]-5-phenylpentanamide | CHEMBL113208 | Statine-like inhibitor A |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H44N6O6 |
|---|
| Mol. Mass. | 596.7177 |
|---|
| SMILES | CC(C)C[C@H](NC(=O)[C@H](C)NC(=O)C[C@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)c1ccccn1)C(C)C)C(N)=O |r| |
|---|
| Structure | 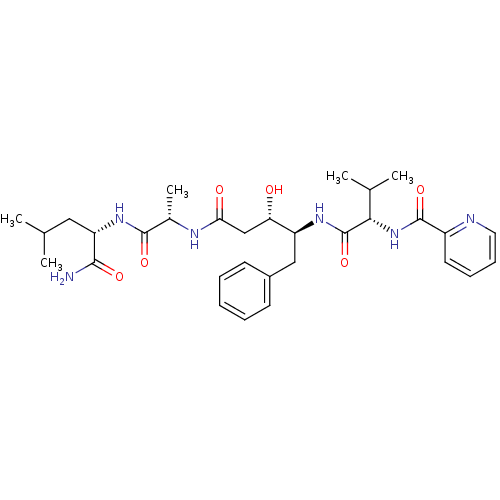
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Leung, D; Abbenante, G; Fairlie, DP Protease inhibitors: current status and future prospects. J Med Chem43:305-41 (2000) [PubMed]
Leung, D; Abbenante, G; Fairlie, DP Protease inhibitors: current status and future prospects. J Med Chem43:305-41 (2000) [PubMed]