| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM17278 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48316 (CHEMBL658079) |
|---|
| Ki | 22000±n/a nM |
|---|
| Citation |  Phillips, G; Guilford, WJ; Buckman, BO; Davey, DD; Eagen, KA; Koovakkat, S; Liang, A; McCarrick, M; Mohan, R; Ng, HP; Pinkerton, M; Subramanyam, B; Ho, E; Trinh, L; Whitlow, M; Wu, S; Xu, W; Morrissey, MM Design, synthesis, and activity of a novel series of factor Xa inhibitors: optimization of arylamidine groups. J Med Chem45:2484-93 (2002) [PubMed] Phillips, G; Guilford, WJ; Buckman, BO; Davey, DD; Eagen, KA; Koovakkat, S; Liang, A; McCarrick, M; Mohan, R; Ng, HP; Pinkerton, M; Subramanyam, B; Ho, E; Trinh, L; Whitlow, M; Wu, S; Xu, W; Morrissey, MM Design, synthesis, and activity of a novel series of factor Xa inhibitors: optimization of arylamidine groups. J Med Chem45:2484-93 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM17278 |
|---|
| n/a |
|---|
| Name | BDBM17278 |
|---|
| Synonyms: | 3-{[6-(3-carbamimidoylphenoxy)-3,5-difluoro-4-methylpyridin-2-yl]oxy}benzene-1-carboximidamide | CHEMBL49636 | Pyridine template, III | ZK-805623 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H17F2N5O2 |
|---|
| Mol. Mass. | 397.3781 |
|---|
| SMILES | Cc1c(F)c(Oc2cccc(c2)C(N)=N)nc(Oc2cccc(c2)C(N)=N)c1F |
|---|
| Structure | 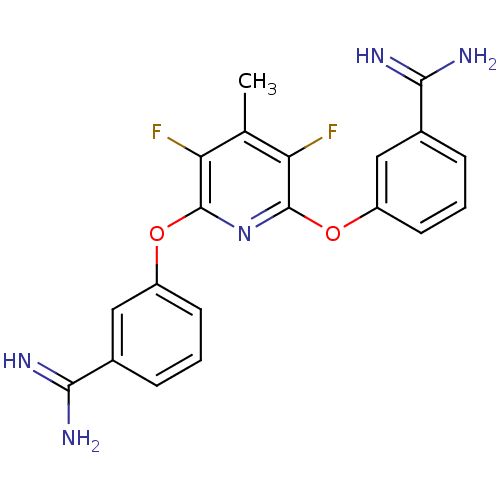
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Phillips, G; Guilford, WJ; Buckman, BO; Davey, DD; Eagen, KA; Koovakkat, S; Liang, A; McCarrick, M; Mohan, R; Ng, HP; Pinkerton, M; Subramanyam, B; Ho, E; Trinh, L; Whitlow, M; Wu, S; Xu, W; Morrissey, MM Design, synthesis, and activity of a novel series of factor Xa inhibitors: optimization of arylamidine groups. J Med Chem45:2484-93 (2002) [PubMed]
Phillips, G; Guilford, WJ; Buckman, BO; Davey, DD; Eagen, KA; Koovakkat, S; Liang, A; McCarrick, M; Mohan, R; Ng, HP; Pinkerton, M; Subramanyam, B; Ho, E; Trinh, L; Whitlow, M; Wu, S; Xu, W; Morrissey, MM Design, synthesis, and activity of a novel series of factor Xa inhibitors: optimization of arylamidine groups. J Med Chem45:2484-93 (2002) [PubMed]