| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carbonic anhydrase 2 |
|---|
| Ligand | BDBM50228328 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_45044 (CHEMBL658042) |
|---|
| Ki | 33±n/a nM |
|---|
| Citation |  Scozzafava, A; Menabuoni, L; Mincione, F; Supuran, CT Carbonic anhydrase inhibitors. A general approach for the preparation of water-soluble sulfonamides incorporating polyamino-polycarboxylate tails and of their metal complexes possessing long-lasting, topical intraocular pressure-lowering properties. J Med Chem45:1466-76 (2002) [PubMed] Scozzafava, A; Menabuoni, L; Mincione, F; Supuran, CT Carbonic anhydrase inhibitors. A general approach for the preparation of water-soluble sulfonamides incorporating polyamino-polycarboxylate tails and of their metal complexes possessing long-lasting, topical intraocular pressure-lowering properties. J Med Chem45:1466-76 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carbonic anhydrase 2 |
|---|
| Name: | Carbonic anhydrase 2 |
|---|
| Synonyms: | CA-II | CA2 | CAC | CAH2_HUMAN | Carbonate dehydratase II | Carbonic anhydrase 2 (CA II) | Carbonic anhydrase 2 (CA-II) | Carbonic anhydrase 2 (Recombinant CA II) | Carbonic anhydrase C | Carbonic anhydrase II (CA II) | Carbonic anhydrase II (CA-II) | Carbonic anhydrase II (CAII) | Carbonic anhydrase II (hCA II) | Carbonic anhydrase isoenzyme II (hCA II) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 29250.71 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00918 |
|---|
| Residue: | 260 |
|---|
| Sequence: | MSHHWGYGKHNGPEHWHKDFPIAKGERQSPVDIDTHTAKYDPSLKPLSVSYDQATSLRIL
NNGHAFNVEFDDSQDKAVLKGGPLDGTYRLIQFHFHWGSLDGQGSEHTVDKKKYAAELHL
VHWNTKYGDFGKAVQQPDGLAVLGIFLKVGSAKPGLQKVVDVLDSIKTKGKSADFTNFDP
RGLLPESLDYWTYPGSLTTPPLLECVTWIVLKEPISVSSEQVLKFRKLNFNGEGEPEELM
VDNWRPAQPLKNRQIKASFK
|
|
|
|---|
| BDBM50228328 |
|---|
| n/a |
|---|
| Name | BDBM50228328 |
|---|
| Synonyms: | ([2-(Carboxymethyl-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-ethyl]-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-acetic acid; compound with Zn complex | ([2-(carboxymethyl-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-ethyl]-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-acetic acid | CHEMBL1795054 | CHEMBL34526 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H36N6O10S2 |
|---|
| Mol. Mass. | 656.728 |
|---|
| SMILES | NS(=O)(=O)c1ccc(CCNC(=O)CN(CCN(CC(O)=O)CC(=O)NCCc2ccc(cc2)S(N)(=O)=O)CC(O)=O)cc1 |
|---|
| Structure | 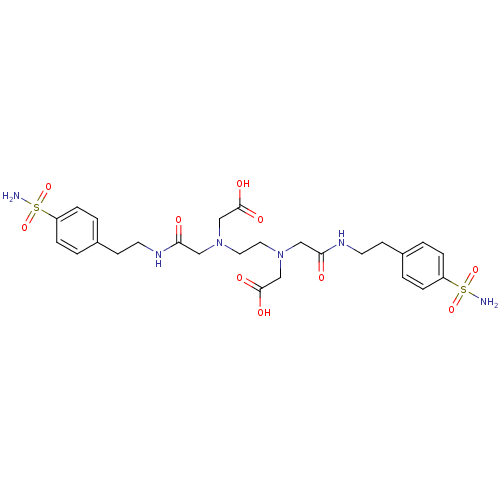
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Scozzafava, A; Menabuoni, L; Mincione, F; Supuran, CT Carbonic anhydrase inhibitors. A general approach for the preparation of water-soluble sulfonamides incorporating polyamino-polycarboxylate tails and of their metal complexes possessing long-lasting, topical intraocular pressure-lowering properties. J Med Chem45:1466-76 (2002) [PubMed]
Scozzafava, A; Menabuoni, L; Mincione, F; Supuran, CT Carbonic anhydrase inhibitors. A general approach for the preparation of water-soluble sulfonamides incorporating polyamino-polycarboxylate tails and of their metal complexes possessing long-lasting, topical intraocular pressure-lowering properties. J Med Chem45:1466-76 (2002) [PubMed]