| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM50122816 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2813 (CHEMBL617842) |
|---|
| Ki | 140±n/a nM |
|---|
| Citation |  Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT2C | 5-hydroxytryptamine receptor 2C | 5-hydroxytryptamine receptor 2C (5HT2c) | 5HT2C_RAT | 5ht1c | Htr1c | Htr2c | Serotonin (5-HT) receptor | Serotonin receptor 2a and 2c (5HT2A and 5HT2C) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51935.10 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | P08909 |
|---|
| Residue: | 460 |
|---|
| Sequence: | MVNLGNAVRSLLMHLIGLLVWQFDISISPVAAIVTDTFNSSDGGRLFQFPDGVQNWPALS
IVVIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYV
WPLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVW
AISIGVSVPIPVIGLRDESKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYFLTIY
VLRRQTLMLLRGHTEEELANMSLNFLNCCCKKNGGEEENAPNPNPDQKPRRKKKEKRPRG
TMQAINNEKKASKVLGIVFFVFLIMWCPFFITNILSVLCGKACNQKLMEKLLNVFVWIGY
VCSGINPLVYTLFNKIYRRAFSKYLRCDYKPDKKPPVRQIPRVAATALSGRELNVNIYRH
TNERVARKANDPEPGIEMQVENLELPVNPSNVVSERISSV
|
|
|
|---|
| BDBM50122816 |
|---|
| n/a |
|---|
| Name | BDBM50122816 |
|---|
| Synonyms: | 1-(2-{4-[1-(4-Fluoro-phenyl)-5-(3-methyl-3H-[1,2,3]triazol-4-yl)-1H-indol-3-yl]-piperidin-1-yl}-ethyl)-imidazolidin-2-one | CHEMBL97184 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H30FN7O |
|---|
| Mol. Mass. | 487.5718 |
|---|
| SMILES | Cn1nncc1-c1ccc2n(cc(C3CCN(CCN4CCNC4=O)CC3)c2c1)-c1ccc(F)cc1 |
|---|
| Structure | 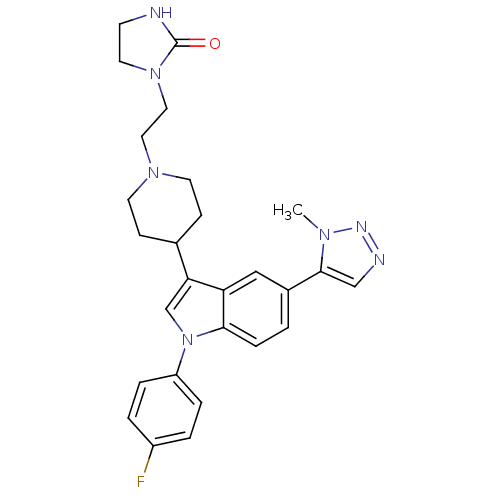
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article
Balle, T; Perregaard, J; Ramirez, MT; Larsen, AK; Søby, KK; Liljefors, T; Andersen, K Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective alpha(1) adrenoceptor antagonists. J Med Chem46:265-83 (2003) [PubMed] Article