| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM50166901 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_303671 (CHEMBL830432) |
|---|
| Ki | 1.2±n/a nM |
|---|
| Citation |  Childers, WE; Abou-Gharbia, MA; Kelly, MG; Andree, TH; Harrison, BL; Ho, DM; Hornby, G; Huryn, DM; Potestio, L; Rosenzweig-Lipson, SJ; Schmid, J; Smith, DL; Sukoff, SJ; Zhang, G; Schechter, LE Synthesis and biological evaluation of benzodioxanylpiperazine derivatives as potent serotonin 5-HT(1A) antagonists: the discovery of Lecozotan. J Med Chem48:3467-70 (2005) [PubMed] Article Childers, WE; Abou-Gharbia, MA; Kelly, MG; Andree, TH; Harrison, BL; Ho, DM; Hornby, G; Huryn, DM; Potestio, L; Rosenzweig-Lipson, SJ; Schmid, J; Smith, DL; Sukoff, SJ; Zhang, G; Schechter, LE Synthesis and biological evaluation of benzodioxanylpiperazine derivatives as potent serotonin 5-HT(1A) antagonists: the discovery of Lecozotan. J Med Chem48:3467-70 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 46122.49 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVLSPGQGNNTTSPPAPFETGGNTTGISDVTVSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVKKVEKTGADT
RHGASPAPQPKKSVNGESGSRNWRLGVESKAGGALCANGAVRQGDDGAALEVIEVHRVGN
SKEHLPLPSEAGPTPCAPASFERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPTLLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RQ
|
|
|
|---|
| BDBM50166901 |
|---|
| n/a |
|---|
| Name | BDBM50166901 |
|---|
| Synonyms: | 4-Chloro-N-{(R)-2-[4-(2,3-dihydro-benzo[1,4]dioxin-5-yl)-piperazin-1-yl]-propyl}-N-pyridin-2-yl-benzamide | CHEMBL371149 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H29ClN4O3 |
|---|
| Mol. Mass. | 492.997 |
|---|
| SMILES | C[C@H](CN(C(=O)c1ccc(Cl)cc1)c1ccccn1)N1CCN(CC1)c1cccc2OCCOc12 |
|---|
| Structure | 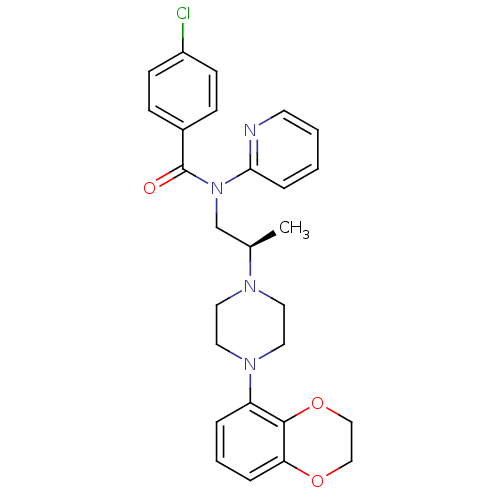
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Childers, WE; Abou-Gharbia, MA; Kelly, MG; Andree, TH; Harrison, BL; Ho, DM; Hornby, G; Huryn, DM; Potestio, L; Rosenzweig-Lipson, SJ; Schmid, J; Smith, DL; Sukoff, SJ; Zhang, G; Schechter, LE Synthesis and biological evaluation of benzodioxanylpiperazine derivatives as potent serotonin 5-HT(1A) antagonists: the discovery of Lecozotan. J Med Chem48:3467-70 (2005) [PubMed] Article
Childers, WE; Abou-Gharbia, MA; Kelly, MG; Andree, TH; Harrison, BL; Ho, DM; Hornby, G; Huryn, DM; Potestio, L; Rosenzweig-Lipson, SJ; Schmid, J; Smith, DL; Sukoff, SJ; Zhang, G; Schechter, LE Synthesis and biological evaluation of benzodioxanylpiperazine derivatives as potent serotonin 5-HT(1A) antagonists: the discovery of Lecozotan. J Med Chem48:3467-70 (2005) [PubMed] Article