| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasminogen |
|---|
| Ligand | BDBM50068488 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302281 (CHEMBL830299) |
|---|
| Ki | 23000±n/a nM |
|---|
| Citation |  Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Plasminogen |
|---|
| Name: | Plasminogen |
|---|
| Synonyms: | Activation peptide | Angiostatin | PLG | PLMN_HUMAN | Plasmin | Plasmin heavy chain A | Plasmin heavy chain A, short form | Plasmin light chain B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 90579.18 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 810 |
|---|
| Sequence: | MEHKEVVLLLLLFLKSGQGEPLDDYVNTQGASLFSVTKKQLGAGSIEECAAKCEEDEEFT
CRAFQYHSKEQQCVIMAENRKSSIIIRMRDVVLFEKKVYLSECKTGNGKNYRGTMSKTKN
GITCQKWSSTSPHRPRFSPATHPSEGLEENYCRNPDNDPQGPWCYTTDPEKRYDYCDILE
CEEECMHCSGENYDGKISKTMSGLECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRE
LRPWCFTTDPNKRWELCDIPRCTTPPPSSGPTYQCLKGTGENYRGNVAVTVSGHTCQHWS
AQTPHTHNRTPENFPCKNLDENYCRNPDGKRAPWCHTTNSQVRWEYCKIPSCDSSPVSTE
QLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRHQKTPENYPNAG
LTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKCSGTEASVVAPPPVVLLPDVETPSEED
CMFGNGKGYRGKRATTVTGTPCQDWAAQEPHRHSIFTPETNPRAGLEKNYCRNPDGDVGG
PWCYTTNPRKLYDYCDVPQCAAPSFDCGKPQVEPKKCPGRVVGGCVAHPHSWPWQVSLRT
RFGMHFCGGTLISPEWVLTAAHCLEKSPRPSSYKVILGAHQEVNLEPHVQEIEVSRLFLE
PTRKDIALLKLSSPAVITDKVIPACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQ
LPVIENKVCNRYEFLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSW
GLGCARPNKPGVYVRVSRFVTWIEGVMRNN
|
|
|
|---|
| BDBM50068488 |
|---|
| n/a |
|---|
| Name | BDBM50068488 |
|---|
| Synonyms: | (S)-3-(7-Carbamimidoyl-naphthalen-2-yl)-2-{4-[(S)-1-(1-imino-ethyl)-pyrrolidin-3-yloxy]-phenyl}-propionic acid; hydrochloride | CHEMBL539053 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H28N4O3 |
|---|
| Mol. Mass. | 444.5255 |
|---|
| SMILES | CC(=N)N1CC[C@@H](C1)Oc1ccc(cc1)[C@H](Cc1ccc2ccc(cc2c1)C(N)=N)C(O)=O |
|---|
| Structure | 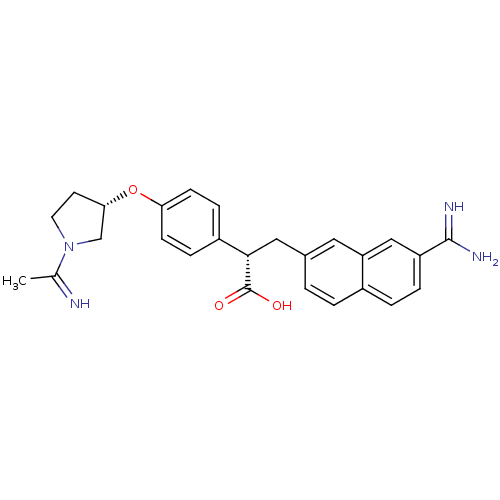
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article
Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article