| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Somatostatin receptor type 5 |
|---|
| Ligand | BDBM50165174 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_306868 (CHEMBL827603) |
|---|
| IC50 | 110±n/a nM |
|---|
| Citation |  Schottelius, M; Reubi, JC; Eltschinger, V; Schwaiger, M; Wester, HJ N-terminal sugar conjugation and C-terminal Thr-for-Thr(ol) exchange in radioiodinated Tyr3-octreotide: effect on cellular ligand trafficking in vitro and tumor accumulation in vivo. J Med Chem48:2778-89 (2005) [PubMed] Article Schottelius, M; Reubi, JC; Eltschinger, V; Schwaiger, M; Wester, HJ N-terminal sugar conjugation and C-terminal Thr-for-Thr(ol) exchange in radioiodinated Tyr3-octreotide: effect on cellular ligand trafficking in vitro and tumor accumulation in vivo. J Med Chem48:2778-89 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Somatostatin receptor type 5 |
|---|
| Name: | Somatostatin receptor type 5 |
|---|
| Synonyms: | SOMATOSTATIN SST5 | SS-5-R | SS5-R | SS5R | SSR5_HUMAN | SSTR5 | Somatostatin receptor type 5 (SSTR5) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 39218.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35346 |
|---|
| Residue: | 364 |
|---|
| Sequence: | MEPLFPASTPSWNASSPGAASGGGDNRTLVGPAPSAGARAVLVPVLYLLVCAAGLGGNTL
VIYVVLRFAKMKTVTNIYILNLAVADVLYMLGLPFLATQNAASFWPFGPVLCRLVMTLDG
VNQFTSVFCLTVMSVDRYLAVVHPLSSARWRRPRVAKLASAAAWVLSLCMSLPLLVFADV
QEGGTCNASWPEPVGLWGAVFIIYTAVLGFFAPLLVICLCYLLIVVKVRAAGVRVGCVRR
RSERKVTRMVLVVVLVFAGCWLPFFTVNIVNLAVALPQEPASAGLYFFVVILSYANSCAN
PVLYGFLSDNFRQSFQKVLCLRKGSGAKDADATEPRPDRIRQQQEATPPAHRAAANGLMQ
TSKL
|
|
|
|---|
| BDBM50165174 |
|---|
| n/a |
|---|
| Name | BDBM50165174 |
|---|
| Synonyms: | 4-[(4R,7S,10S,13R,16S)-19-[2-({[(4R)-4-{[(2S,4R,5S)-3,4-dihydroxy-6-(hydroxymethyl)-5-{[(2S,4S,5S)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxan-2-yl]oxy}-2,3,5-trihydroxyoxan-2-yl]methyl}azaniumyl)-3-phenylpropanamido]-4-{[(2R)-1,3-dihydroxybutan-2-yl]carbamoyl}-16-[(4-hydroxy-3-iodophenyl)methyl]-7-(1-hydroxyethyl)-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentaazacycloicosan-10-yl]butan-1-aminium | Radiolabeled octreotide derivative |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C67H97IN10O26S2 |
|---|
| Mol. Mass. | 1649.572 |
|---|
| SMILES | CC(O)[C@@H](CO)NC(=O)[C@@H]1CSSCC(NC(=O)C(Cc2ccccc2)[NH2+]CC2(O)OCC(O)[C@@H](O[C@@H]3OC(CO)[C@@H](O[C@@H]4OC(CO)[C@@H](O)[C@H](O)C4O)[C@H](O)C3O)C2O)C(=O)N[C@@H](Cc2ccc(O)c(I)c2)C(=O)N[C@H](Cc2c[nH]c3ccccc23)C(=O)N[C@@H](CCCC[NH3+])C(=O)N[C@@H](C(C)O)C(=O)N1 |
|---|
| Structure | 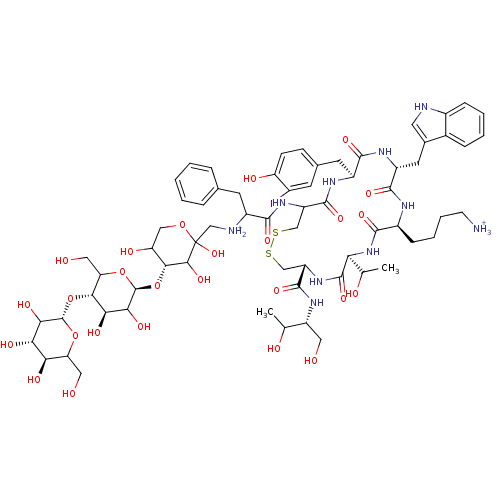
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Schottelius, M; Reubi, JC; Eltschinger, V; Schwaiger, M; Wester, HJ N-terminal sugar conjugation and C-terminal Thr-for-Thr(ol) exchange in radioiodinated Tyr3-octreotide: effect on cellular ligand trafficking in vitro and tumor accumulation in vivo. J Med Chem48:2778-89 (2005) [PubMed] Article
Schottelius, M; Reubi, JC; Eltschinger, V; Schwaiger, M; Wester, HJ N-terminal sugar conjugation and C-terminal Thr-for-Thr(ol) exchange in radioiodinated Tyr3-octreotide: effect on cellular ligand trafficking in vitro and tumor accumulation in vivo. J Med Chem48:2778-89 (2005) [PubMed] Article