| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2X purinoceptor 1 |
|---|
| Ligand | BDBM50118221 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_321555 (CHEMBL881969) |
|---|
| IC50 | 14±n/a nM |
|---|
| Citation |  Minetti, P; Tinti, MO; Carminati, P; Castorina, M; Di Cesare, MA; Di Serio, S; Gallo, G; Ghirardi, O; Giorgi, F; Giorgi, L; Piersanti, G; Bartoccini, F; Tarzia, G 2-n-Butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine and analogues as A2A adenosine receptor antagonists. Design, synthesis, and pharmacological characterization. J Med Chem48:6887-96 (2005) [PubMed] Article Minetti, P; Tinti, MO; Carminati, P; Castorina, M; Di Cesare, MA; Di Serio, S; Gallo, G; Ghirardi, O; Giorgi, F; Giorgi, L; Piersanti, G; Bartoccini, F; Tarzia, G 2-n-Butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine and analogues as A2A adenosine receptor antagonists. Design, synthesis, and pharmacological characterization. J Med Chem48:6887-96 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2X purinoceptor 1 |
|---|
| Name: | P2X purinoceptor 1 |
|---|
| Synonyms: | ATP receptor | P2RX1_RAT | P2X1 | P2rx1 | Purinergic receptor | Purinergic, P2X1 | RP-2 protein |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 44969.95 |
|---|
| Organism: | RAT |
|---|
| Description: | Purinergic, P2X1 0 RAT::P47824 |
|---|
| Residue: | 399 |
|---|
| Sequence: | MARRLQDELSAFFFEYDTPRMVLVRNKKVGVIFRLIQLVVLVYVIGWVFVYEKGYQTSSD
LISSVSVKLKGLAVTQLQGLGPQVWDVADYVFPAHGDSSFVVMTNFIVTPQQTQGHCAEN
PEGGICQDDSGCTPGKAERKAQGIRTGNCVPFNGTVKTCEIFGWCPVEVDDKIPSPALLR
EAENFTLFIKNSISFPRFKVNRRNLVEEVNGTYMKKCLYHKIQHPLCPVFNLGYVVRESG
QDFRSLAEKGGVVGITIDWKCDLDWHVRHCKPIYQFHGLYGEKNLSPGFNFRFARHFVQN
GTNRRHLFKVFGIHFDILVDGKAGKFDIIPTMTTIGSGIGIFGVATVLCDLLLLHILPKR
HYYKQKKFKYAEDMGPGEGEHDPVATSSTLGLQENMRTS
|
|
|
|---|
| BDBM50118221 |
|---|
| n/a |
|---|
| Name | BDBM50118221 |
|---|
| Synonyms: | 9H-purine derivative | CHEMBL132722 | DIPHOSPHOMETHYLPHOSPHONIC ACID ADENOSYL ESTER | alpha,beta-meATP |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H18N5O12P3 |
|---|
| Mol. Mass. | 505.2082 |
|---|
| SMILES | Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |
|---|
| Structure | 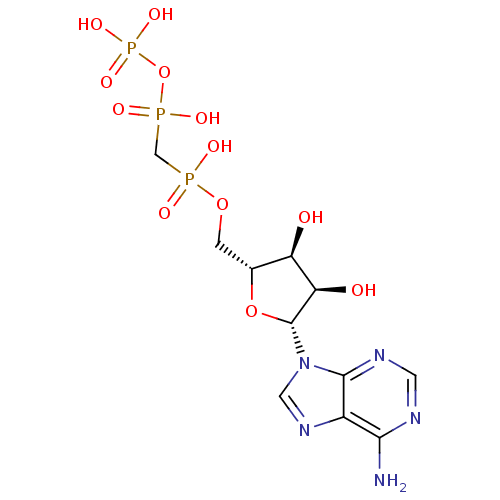
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Minetti, P; Tinti, MO; Carminati, P; Castorina, M; Di Cesare, MA; Di Serio, S; Gallo, G; Ghirardi, O; Giorgi, F; Giorgi, L; Piersanti, G; Bartoccini, F; Tarzia, G 2-n-Butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine and analogues as A2A adenosine receptor antagonists. Design, synthesis, and pharmacological characterization. J Med Chem48:6887-96 (2005) [PubMed] Article
Minetti, P; Tinti, MO; Carminati, P; Castorina, M; Di Cesare, MA; Di Serio, S; Gallo, G; Ghirardi, O; Giorgi, F; Giorgi, L; Piersanti, G; Bartoccini, F; Tarzia, G 2-n-Butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine and analogues as A2A adenosine receptor antagonists. Design, synthesis, and pharmacological characterization. J Med Chem48:6887-96 (2005) [PubMed] Article