| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Oxytocin receptor |
|---|
| Ligand | BDBM50326722 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_326541 (CHEMBL863361) |
|---|
| Ki | 0.67±n/a nM |
|---|
| Citation |  Quattropani, A; Dorbais, J; Covini, D; Pittet, PA; Colovray, V; Thomas, RJ; Coxhead, R; Halazy, S; Scheer, A; Missotten, M; Ayala, G; Bradshaw, C; De Raemy-Schenk, AM; Nichols, A; Cirillo, R; Tos, EG; Giachetti, C; Golzio, L; Marinelli, P; Church, DJ; Barberis, C; Chollet, A; Schwarz, MK Discovery and development of a new class of potent, selective, orally active oxytocin receptor antagonists. J Med Chem48:7882-905 (2005) [PubMed] Article Quattropani, A; Dorbais, J; Covini, D; Pittet, PA; Colovray, V; Thomas, RJ; Coxhead, R; Halazy, S; Scheer, A; Missotten, M; Ayala, G; Bradshaw, C; De Raemy-Schenk, AM; Nichols, A; Cirillo, R; Tos, EG; Giachetti, C; Golzio, L; Marinelli, P; Church, DJ; Barberis, C; Chollet, A; Schwarz, MK Discovery and development of a new class of potent, selective, orally active oxytocin receptor antagonists. J Med Chem48:7882-905 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Oxytocin receptor |
|---|
| Name: | Oxytocin receptor |
|---|
| Synonyms: | OT-R | OXYR_RAT | Otr | Oxtr | Oxytocin |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 42889.28 |
|---|
| Organism: | RAT |
|---|
| Description: | Oxytocin OXTR RAT::P70536 |
|---|
| Residue: | 388 |
|---|
| Sequence: | MEGTPAANWSVELDLGSGVPPGEEGNRTAGPPQRNEALARVEVAVLCLILFLALSGNACV
LLALRTTRHKHSRLFFFMKHLSIADLVVAVFQVLPQLLWDITFRFYGPDLLCRLVKYLQV
VGMFASTYLLLLMSLDRCLAICQPLRSLRRRTDRLAVLGTWLGCLVASAPQVHIFSLREV
ADGVFDCWAVFIQPWGPKAYVTWITLAVYIVPVIVLAACYGLISFKIWQNLRLKTAAAAA
AAEGNDAAGGAGRAALARVSSVKLISKAKIRTVKMTFIIVLAFIVCWTPFFFVQMWSVWD
VNAPKEASAFIIAMLLASLNSCCNPWIYMLFTGHLFHELVQRFFCCSARYLKGSRPGETS
VSKKSNSSTFVLSRRSSSQRSCSQPSSA
|
|
|
|---|
| BDBM50326722 |
|---|
| n/a |
|---|
| Name | BDBM50326722 |
|---|
| Synonyms: | (Z)-3-(4-(N-(4-chlorophenyl)-N-(2-oxo-2-(2-(2-oxoindolin-3-ylidene)hydrazinyl)ethyl)sulfamoyl)phenyl)-N-(3-(dimethylamino)propyl)propanamide | 3-(4-(N-(4-chlorophenyl)-N-(2-oxo-2-(2-(2-oxoindolin-3-ylidene)hydrazinyl)ethyl)sulfamoyl)phenyl)-N-(3-(dimethylamino)propyl)propanamide | CHEMBL445816 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H33ClN6O5S |
|---|
| Mol. Mass. | 625.138 |
|---|
| SMILES | CN(C)CCCNC(=O)CCc1ccc(cc1)S(=O)(=O)N(CC(=O)N\N=C1/C(=O)Nc2ccccc12)c1ccc(Cl)cc1 |
|---|
| Structure | 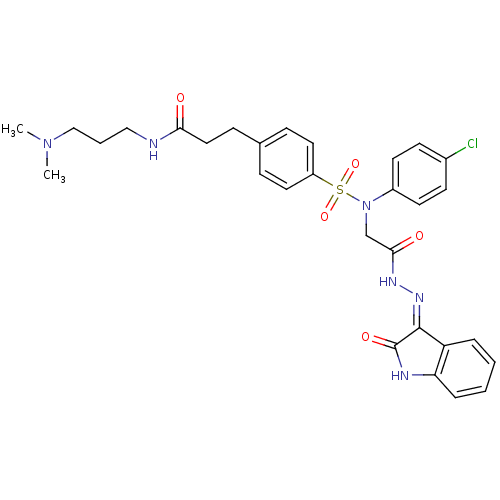
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Quattropani, A; Dorbais, J; Covini, D; Pittet, PA; Colovray, V; Thomas, RJ; Coxhead, R; Halazy, S; Scheer, A; Missotten, M; Ayala, G; Bradshaw, C; De Raemy-Schenk, AM; Nichols, A; Cirillo, R; Tos, EG; Giachetti, C; Golzio, L; Marinelli, P; Church, DJ; Barberis, C; Chollet, A; Schwarz, MK Discovery and development of a new class of potent, selective, orally active oxytocin receptor antagonists. J Med Chem48:7882-905 (2005) [PubMed] Article
Quattropani, A; Dorbais, J; Covini, D; Pittet, PA; Colovray, V; Thomas, RJ; Coxhead, R; Halazy, S; Scheer, A; Missotten, M; Ayala, G; Bradshaw, C; De Raemy-Schenk, AM; Nichols, A; Cirillo, R; Tos, EG; Giachetti, C; Golzio, L; Marinelli, P; Church, DJ; Barberis, C; Chollet, A; Schwarz, MK Discovery and development of a new class of potent, selective, orally active oxytocin receptor antagonists. J Med Chem48:7882-905 (2005) [PubMed] Article