| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carboxylic ester hydrolase |
|---|
| Ligand | BDBM50211243 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_455572 (CHEMBL886352) |
|---|
| IC50 | 1950±n/a nM |
|---|
| Citation |  Tang, H; Ning, FX; Wei, YB; Huang, SL; Huang, ZS; Chan, AS; Gu, LQ Derivatives of oxoisoaporphine alkaloids: a novel class of selective acetylcholinesterase inhibitors. Bioorg Med Chem Lett17:3765-8 (2007) [PubMed] Article Tang, H; Ning, FX; Wei, YB; Huang, SL; Huang, ZS; Chan, AS; Gu, LQ Derivatives of oxoisoaporphine alkaloids: a novel class of selective acetylcholinesterase inhibitors. Bioorg Med Chem Lett17:3765-8 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carboxylic ester hydrolase |
|---|
| Name: | Carboxylic ester hydrolase |
|---|
| Synonyms: | BuChE | Butyrlcholinesterase (BuChE) | Butyrylcholine esterase | Butyrylcholinesterase | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | Butyrylcholinesterase (EqBuChE) | Carboxylic ester hydrolase | butyrylcholinesterase precursor |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 68842.83 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | Q9N1N9 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MQSWGTIICIRILLRFLLLWVLIGNSHTEEDIIITTKNGKVRGMNLPVLGGTVTAFLGIP
YAQPPLGRLRFKKPQSLTKWSNIWNATKYANSCYQNTDQSFPGFLGSEMWNPNTELSEDC
LYLNVWIPAPKPKNATVMIWIYGGGFQTGTSSLPVYDGKFLARVERVIVVSMNYRVGALG
FLALSENPEAPGNMGLFDQQLALQWVQKNIAAFGGNPRSVTLFGESAGAASVSLHLLSPR
SQPLFTRAILQSGSSNAPWAVTSLYEARNRTLTLAKRMGCSRDNETEMIKCLRDKDPQEI
LLNEVFVVPYDTLLSVNFGPTVDGDFLTDMPDTLLQLGQFKRTQILVGVNKDEGTAFLVY
GAPGFSKDNNSIITRKEFQEGLKIFFPRVSEFGRESILFHYMDWLDDQRAENYREALDDV
VGDYNIICPALEFTKKFSELGNDAFFYYFEHRSTKLPWPEWMGVMHGYEIEFVFGLPLER
RVNYTKAEEILSRSIMKRWANFAKYGNPNGTQSNSTRWPVFKSTEQKYLTLNTESPKVYT
KLRAQQCRFWTLFFPKVLELTGNIDEAEREWKAGFHRWNNYMMDWKNQFNDYTSKKESCS
DF
|
|
|
|---|
| BDBM50211243 |
|---|
| n/a |
|---|
| Name | BDBM50211243 |
|---|
| Synonyms: | 3-dimethylamino-N-(7-oxo-7H-1-aza-benzo[de]anthracen-9-yl)-propionamide | 9-[3-(Dimethylamino)propionamido]-1-azabenzanthrone | CHEMBL245471 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H19N3O2 |
|---|
| Mol. Mass. | 345.3945 |
|---|
| SMILES | CN(C)CCC(=O)Nc1ccc-2c(c1)C(=O)c1cccc3ccnc-2c13 |
|---|
| Structure | 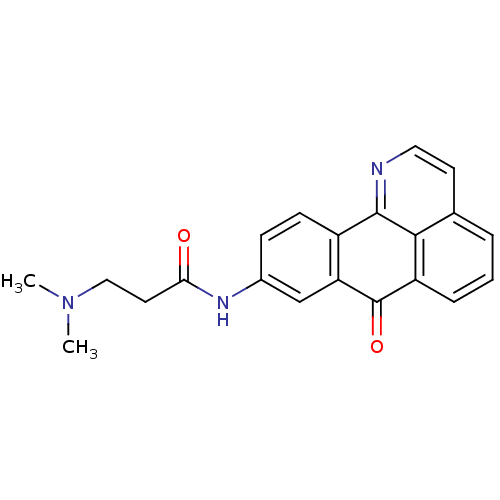
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tang, H; Ning, FX; Wei, YB; Huang, SL; Huang, ZS; Chan, AS; Gu, LQ Derivatives of oxoisoaporphine alkaloids: a novel class of selective acetylcholinesterase inhibitors. Bioorg Med Chem Lett17:3765-8 (2007) [PubMed] Article
Tang, H; Ning, FX; Wei, YB; Huang, SL; Huang, ZS; Chan, AS; Gu, LQ Derivatives of oxoisoaporphine alkaloids: a novel class of selective acetylcholinesterase inhibitors. Bioorg Med Chem Lett17:3765-8 (2007) [PubMed] Article