| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glucocorticoid receptor |
|---|
| Ligand | BDBM50221153 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_457479 (CHEMBL941997) |
|---|
| Ki | 200±n/a nM |
|---|
| Citation |  Clark, RD; Ray, NC; Blaney, P; Crackett, PH; Hurley, C; Williams, K; Dyke, HJ; Clark, DE; Lockey, PM; Devos, R; Wong, M; White, A; Belanoff, JK 2-Benzenesulfonyl-8a-benzyl-hexahydro-2H-isoquinolin-6-ones as selective glucocorticoid receptor antagonists. Bioorg Med Chem Lett17:5704-8 (2007) [PubMed] Article Clark, RD; Ray, NC; Blaney, P; Crackett, PH; Hurley, C; Williams, K; Dyke, HJ; Clark, DE; Lockey, PM; Devos, R; Wong, M; White, A; Belanoff, JK 2-Benzenesulfonyl-8a-benzyl-hexahydro-2H-isoquinolin-6-ones as selective glucocorticoid receptor antagonists. Bioorg Med Chem Lett17:5704-8 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glucocorticoid receptor |
|---|
| Name: | Glucocorticoid receptor |
|---|
| Synonyms: | GCR_HUMAN | GR | GRL | Glucocorticoid | Glucocorticoid receptor (GRFP) | NR3C1 | Nuclear receptor subfamily 3 group C member 1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 85656.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04150 |
|---|
| Residue: | 777 |
|---|
| Sequence: | MDSKESLTPGREENPSSVLAQERGDVMDFYKTLRGGATVKVSASSPSLAVASQSDSKQRR
LLVDFPKGSVSNAQQPDLSKAVSLSMGLYMGETETKVMGNDLGFPQQGQISLSSGETDLK
LLEESIANLNRSTSVPENPKSSASTAVSAAPTEKEFPKTHSDVSSEQQHLKGQTGTNGGN
VKLYTTDQSTFDILQDLEFSSGSPGKETNESPWRSDLLIDENCLLSPLAGEDDSFLLEGN
SNEDCKPLILPDTKPKIKDNGDLVLSSPSNVTLPQVKTEKEDFIELCTPGVIKQEKLGTV
YCQASFPGANIIGNKMSAISVHGVSTSGGQMYHYDMNTASLSQQQDQKPIFNVIPPIPVG
SENWNRCQGSGDDNLTSLGTLNFPGRTVFSNGYSSPSMRPDVSSPPSSSSTATTGPPPKL
CLVCSDEASGCHYGVLTCGSCKVFFKRAVEGQHNYLCAGRNDCIIDKIRRKNCPACRYRK
CLQAGMNLEARKTKKKIKGIQQATTGVSQETSENPGNKTIVPATLPQLTPTLVSLLEVIE
PEVLYAGYDSSVPDSTWRIMTTLNMLGGRQVIAAVKWAKAIPGFRNLHLDDQMTLLQYSW
MFLMAFALGWRSYRQSSANLLCFAPDLIINEQRMTLPCMYDQCKHMLYVSSELHRLQVSY
EEYLCMKTLLLLSSVPKDGLKSQELFDEIRMTYIKELGKAIVKREGNSSQNWQRFYQLTK
LLDSMHEVVENLLNYCFQTFLDKTMSIEFPEMLAEIITNQIPKYSNGNIKKLLFHQK
|
|
|
|---|
| BDBM50221153 |
|---|
| n/a |
|---|
| Name | BDBM50221153 |
|---|
| Synonyms: | (R)-8a-(4-methoxybenzyl)-2-(4-tert-butylphenylsulfonyl)-1,2,3,4,8,8a-hexahydroisoquinolin-6(7H)-one | CHEMBL250700 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H33NO4S |
|---|
| Mol. Mass. | 467.62 |
|---|
| SMILES | COc1ccc(C[C@]23CCC(=O)C=C2CCN(C3)S(=O)(=O)c2ccc(cc2)C(C)(C)C)cc1 |c:12| |
|---|
| Structure | 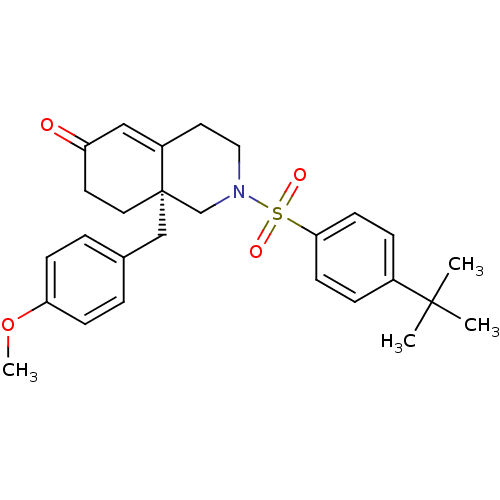
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Clark, RD; Ray, NC; Blaney, P; Crackett, PH; Hurley, C; Williams, K; Dyke, HJ; Clark, DE; Lockey, PM; Devos, R; Wong, M; White, A; Belanoff, JK 2-Benzenesulfonyl-8a-benzyl-hexahydro-2H-isoquinolin-6-ones as selective glucocorticoid receptor antagonists. Bioorg Med Chem Lett17:5704-8 (2007) [PubMed] Article
Clark, RD; Ray, NC; Blaney, P; Crackett, PH; Hurley, C; Williams, K; Dyke, HJ; Clark, DE; Lockey, PM; Devos, R; Wong, M; White, A; Belanoff, JK 2-Benzenesulfonyl-8a-benzyl-hexahydro-2H-isoquinolin-6-ones as selective glucocorticoid receptor antagonists. Bioorg Med Chem Lett17:5704-8 (2007) [PubMed] Article